
4 mg/ml
For the use of a Registered Medical Practitioner or a Hospital or a Institution only.
DEXAMETHASONE SODIUM PHOSPHATE INJECTION USP (Dexamethasone Sodium Phosphate) is a corticosteroid with mainly glucocorticoid activity. Chemically, Dexamethasone Sodium Phosphate is 9-Fluoro-11β,17,21-trihydroxy-16α-methylpregna-1,4-diene-3,20-dione 21-(dihydrogen phosphate) disodium salt. The molecular formula is C22H28FNa2O8P and molecular weight is 516.40.
STRUCTURAL FORMULA :
Its structural formula is :
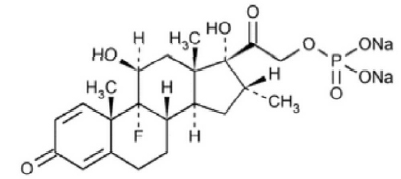
DEXAMETHASONE SODIUM PHOSPHATE INJECTION USP is a sterile, clear, colourless to pale yellow aqueous solution filled in amber ampoule of suitable size.
COMPOSITION :
Each ml contains :
Dexamethasone Sodium Phosphate USP
equivalent to Dexamethasone Phosphate 4 mg
Methylparaben USP 0.15 % w/v
Propylparaben USP 0.02 % w/v
(as preservatives)
Water for Injection USP q.s.
ACTIONS :
Dexamethasone is a synthetic corticosteroid (glucocorticoid). As such its main actions may be grouped as follows :
Anti-inflammatory and Immunological Actions : Glucocorticoids prevent the development of the inflammatory response, i.e. redness, swelling, tenderness. They also inhibit capillary dilation and phagocytosis and appear to prevent the hypersensitivity responses which occur after antigen-antibody reactions. Pharmacological Actions : The principal action of dexamethasone is on gluconeogenesis, glycogen deposition and protein and calcium metabolism, together with inhibition of corticotrophin secretion. Glucocorticoids also influence the mobilisation, oxidation, synthesis and storage of fats. Except for its use in the treatment of adrenal insufficiency it does not cure disease but it is used rather to treat disease symptoms because of its pharmacological properties, i.e. anti-inflammatory and anti-allergic actions.
PHARMACOKINETICS :
Dexamethasone sodium phosphate is absorbed rapidly following intramuscular or intravenous injection. Intramuscular injections of dexamethasone phosphate give maximum plasma concentrations of dexamethasone at one hour. The biological half-life of
dexamethasone is about 190 minutes. In the circulation, small amounts of dexamethasone are bound to plasma proteins. Synthetic corticosteroids such as dexamethasone are less extensively protein bound and more slowly metabolised than hydrocortisone. Dexamethasone penetrates into tissue fluids and cerebrospinal fluids. Metabolism occurs in most tissues, but primarily in the liver. The inactive metabolites are excreted in the urine, mainly as glucuronides and sulfates but also as unconjugated metabolites. Small amounts of unchanged drug are also excreted in the urine. Up to 65 % of a dose of dexamethasone is excreted in the urine within 24 hours.
Administration :
DEXAMETHASONE SODIUM PHOSPHATE INJECTION USP is given by intravenous, intramuscular, intra-articular, intralesional and soft tissue injection. When DEXAMETHASONE SODIUM PHOSPHATE INJECTION USP is given by intravenous infusion, dextrose 5 % in water and sodium chloride 0.9 % have been recommended as diluents. The exact concentration of dexamethasone sodium phosphate per infusion container should be determined by the desired dose, patient fluid intake and drip rate required. The infusion mixture must be used within 24 hours and the usual aseptic techniques for injections should be observed. Solutions used for intravenous administration or further dilution of this product should be preservative-free when used in the neonate, especially the premature infant.
INSTRUCTIONS FOR USE OF AMPOULE :
The ampoule used in this product is equipped with O.P.C. (One Point Cut) opening system. No ampoule file is needed to open the ampoule. The neck of the ampoule is prescored at the point of constriction. A coloured dot on the ampoule head helps to orientate the ampoule. Take the ampoule and face the coloured dot. Let the solution at the head of the ampoule to flow down by shaking or a gentle stroke. The ampoule opens easily by placing the thumb on the coloured dot and gently pressing downwards as shown.

Dosage :
All dosage recommendations are given in units of dexamethasone phosphate.
Intravenous and intramuscular injection :
General considerations :
Dosage must be individualised on the basis of the disease and the response of the patient. In order to minimise side effects, the lowest possible dosage adequate to control the disease process should be used. Usually the parenteral dosage ranges are one-third to one-half of the oral dose, given every 12 hours. The usual initial dosage is 0.5 mg - 20 mg a day. In situations of less severity, lower doses will generally suffice. However, in certain overwhelming, acute, life-threatening situations, administration in dosages exceeding the usual dosage may be justified. In these circumstances, the slower rate of absorption by intramuscular administration should be recognised. Both the dose in the evening, which is useful in alleviating morning stiffness and the divided dosage regimen are associated with greater suppression of the hypothalamo-pituitary-adrenal axis. After a favourable response is noted, the proper maintenance dosage should be determined by decreasing the initial dosage by small amounts at appropriate intervals to the lowest dosage which will maintain an adequate clinical response. Chronic dosage should preferably not exceed 500 micrograms dexamethasone daily. Close monitoring of the drug dosage is needed. To avoid hypoadrenalism and/or a relapse of the underlying disease, it may be necessary to withdraw the drug gradually.
Whenever possible, the intravenous route should be used for the initial dose and for as many subsequent doses as are given while the patient is in shock (because of the irregular rate of absorption of any medicament administered by any other route in such patients). When the blood pressure responds, use the intramuscular route until oral therapy can be substituted. For the comfort of the patient, not more than 2 ml should be injected intramuscularly at any one site. In emergencies, the usual dose of DEXAMETHASONE SODIUM PHOSPHATE INJECTION USP by intravenous or intramuscular injection is 4 mg - 20 mg (in shock use only the intravenous route). This dose may be repeated until adequate response is noted. After initial improvement, single doses of 2 mg - 4 mg, repeated as necessary, should be sufficient. The total daily dosage usually need not exceed 80 mg, even in severe conditions. When constant maximal effect is desired, dosage must be repeated at three-hour or four-hour intervals or maintained by slow intravenous drip. Intravenous or intramuscular injections are advised in acute illness. When the acute stage has passed, oral steroid therapy should be substituted as soon as feasible.
Shock (of haemorrhagic, traumatic, or surgical origin) :
Usually 2 mg - 6 mg/kg body weight as a single intravenous injection. This may be repeated in two to six hours if shock persists. Alternatively, this may be followed immediately by the same dose in an intravenous infusion. Therapy with DEXAMETHASONE SODIUM PHOSPHATE INJECTION USP is an adjunct to and not a replacement for conventional therapy. Administration of these high doses should be continued only until the patient’s condition has stabilised and usually no longer than 48 - 72 hours.
Cerebral oedema :
Associated with primary or metastatic brain tumour, preoperative preparation of patients with increased intracranial pressure secondary to brain tumour : initially 10 mg intravenously, followed by 4 mg intramuscularly every six hours until symptoms of cerebral oedema subside. Response is usually noted within 12 - 24 hours; dosage may be reduced after two to four days and gradually discontinued over five to seven days. High doses of DEXAMETHASONE SODIUM PHOSPHATE INJECTION USP are recommended for initiating short-term intensive therapy for acute life-threatening cerebral oedema. Following the high-loading dose schedule of the first day therapy, the dose is scaled down over the seven- to ten- day period of intensive therapy and subsequently reduced to zero over the next seven to ten days. When maintenance therapy is required, substitute oral dexamethasone phosphate as soon as possible.
CONTRAINDICATIONS :
Unless considered life-saving, systemic administration of DEXAMETHASONE SODIUM PHOSPHATE INJECTION USP is contraindicated in systemic viral and fungal infections and in patients with myasthenia gravis, peptic ulcer, osteoporosis and psychoses.
DEXAMETHASONE SODIUM PHOSPHATE INJECTION USP is also contraindicated in patients who are hypersensitive to the drug or any component of the injection. Administration of live virus vaccines is contraindicated in individuals receiving immunosuppressive doses of corticosteroids.
Because rare instances of anaphylactoid reactions have occurred in patients receiving parenteral corticosteroid therapy, appropriate precautionary measures should be taken prior to administration, especially when the patient has a history of allergy to any drug. Anaphylactoid and hypersensitivity reactions have been reported for DEXAMETHASONE SODIUM PHOSPHATE INJECTION USP. DEXAMETHASONE SODIUM PHOSPHATE INJECTION USP contains sodium metabisulfite, a sulfite that may cause allergic-type reactions including anaphylactic symptoms and life-threatening or less severe asthmatic episodes in certain susceptible people. The overall prevalence of sulfite sensitivity in the general population is unknown and probably low. Sulfite sensitivity is seen more frequently in asthmatic than non-asthmatic people. Corticosteroids may exacerbate systemic fungal infections and therefore should not be used in the presence of such infections unless they are needed to control drug reactions due to amphotericin B. Moreover, there have been cases reported in which concomitant use of amphotericin B and hydrocortisone was followed by cardiac enlargement and congestive failure.
In patients on corticosteroid therapy subjected to any unusual stress, increased dosage of rapidly acting corticosteroids before, during, and after the stressful situation is indicated. Drug-induced secondary adrenocortical insufficiency may result from too rapid withdrawal of corticosteroids and may be minimized by gradual reduction of dosage. This type of relative insufficiency may persist for months after discontinuation of therapy; therefore, in any situation of stress occurring during that period, hormone therapy should be reinstituted. If the patient is receiving steroids already, dosage may have to be increased. Since mineralocorticoid secretion may be impaired, salt and/or a mineralocorticoid should be administered concurrently. Corticosteroids may mask some signs of infection, and new infections may appear during their use. There may be decreased resistance and inability to localize infection when corticosteroids are used. In cerebral malaria, a double-blind trial has shown that the use of corticosteroids is associated with prolongation of coma and a higher incidence of pneumonia and gastrointestinal bleeding.
Corticosteroids may activate latent amebiasis. Therefore, it is recommended that latent or active amebiasis be ruled out before initiating corticosteroid therapy in any patient who has spent time in the tropics or any patient with unexplained diarrhoea. Prolonged use of corticosteroids may produce posterior subcapsular cataracts, glaucoma with possible damage to the optic nerves, and may enhance the establishment of secondary ocular infections due to fungi or viruses. Average and large doses of cortisone or hydrocortisone can cause elevation of blood pressure, salt and water retention, and increased excretion of potassium. These effects are less likely to occur with the synthetic derivatives except when used in large doses. Dietary salt restriction and potassium supplementation may be necessary. All corticosteroids increase calcium excretion. Administration of live virus vaccines, including smallpox, is contraindicated in individuals receiving immunosuppressive doses of corticosteroids. If inactivated viral or bacterial vaccines are administered to individuals receiving immunosuppressive doses of corticosteroids, the expected serum antibody response may not be obtained. However, immunization procedures may be undertaken in patients who are receiving corticosteroids as replacement therapy, e.g., for Addison’s disease.
Patients who are on drugs which suppress the immune system are more susceptible to infections than healthy individuals. Chickenpox and measles, for example, can have a more serious or even fatal course in non-immune patients on corticosteroids. In such patients who have not had these diseases, particular care should be taken to avoid exposure. The risk of developing a disseminated infection varies among individuals and can be related to the dose, route and duration of corticosteroid administration as well as to the underlying disease. If exposed to chickenpox, prophylaxis with varicella zoster immune globulin (VZIG) may be indicated. If chickenpox develops, treatment with antiviral agents may be considered. If exposed to measles, prophylaxis with immune globulin (IG) may be indicated.
Similarly, corticosteroids should be used with great care in patients with known or suspected Strongyloides (threadworm) infestation. In such patients, corticosteroid-induced immunosuppression may lead to Strongyloides hyperinfection and dissemination with widespread larval migration, often accompanied by severe enterocolitis and potentially fatal gram-negative septicaemia.
The use of DEXAMETHASONE SODIUM PHOSPHATE INJECTION USP in active tuberculosis should be restricted to those cases of fulminating or disseminated tuberculosis in which the corticosteroid is used for the management of the disease in conjunction with an appropriate antituberculous regimen. If corticosteroids are indicated in patients with latent tuberculosis or tuberculin reactivity, close observation is necessary as reactivation of the disease may occur. During prolonged corticosteroid therapy, these patients should receive chemoprophylaxis. Literature reports suggest an apparent association between use of corticosteroids and left ventricular free wall rupture after a recent myocardial infarction; therefore, therapy with corticosteroids should be used with great caution in these patients.
PRECAUTIONS :
This product, like many other steroid formulations, is sensitive to heat. Therefore, it should not be autoclaved when it is desirable to sterilize the exterior of the ampoule. Following prolonged therapy, withdrawal of corticosteroids may result in symptoms of the corticosteroid withdrawal syndrome including fever, myalgia, arthralgia, and malaise. This may occur in patients even without evidence of adrenal insufficiency. There is an enhanced effect of corticosteroids in patients with hypothyroidism and in those with cirrhosis. Corticosteroids should be used cautiously in patients with ocular herpes simplex for fear of corneal perforation. The lowest possible dose of corticosteroid should be used to control the condition under treatment, and when reduction in dosage is possible, the reduction must be gradual.
Psychic derangements may appear when corticosteroids are used, ranging from euphoria, insomnia, mood swings, personality changes, and severe depression to frank psychotic manifestations. Also, existing emotional instability or psychotic tendencies may be aggravated by corticosteroids. Steroids should be used with caution in nonspecific ulcerative colitis, if there is a probability of impending perforation, abscess, or other pyogenic infection, also in diverticulitis, fresh intestinal anastomoses, active or latent peptic ulcer, renal insufficiency, hypertension, osteoporosis, and myasthenia gravis. Signs of peritoneal irritation following gastrointestinal perforation in patients receiving large doses of corticosteroids may be minimal or absent. Fat embolism has been reported as a possible complication of hypercortisonism.
When large doses are given, some authorities advise that antacids be administered between meals to help to prevent peptic ulcer. Steroids may increase or decrease motility and number of spermatozoa in some patients. Intra-articular injection of a corticosteroid may produce systemic as well as local effects. Appropriate examination of any joint fluid present is necessary to exclude a septic process. A marked increase in pain accompanied by local swelling, further restriction of joint motion, fever, and malaise is suggestive of
septic arthritis. If this complication occurs and the diagnosis of sepsis is confirmed, appropriate antimicrobial therapy should be instituted. Injection of a steroid into an infected site is to be avoided. Corticosteroids should not be injected into unstable joints. Patients should be impressed strongly with the importance of not overusing joints in which symptomatic benefit has been obtained as long as the inflammatory process remains active. Frequent intra-articular injection may result in damage to joint tissues. The slower rate of absorption by intramuscular administration should be recognized. DEXAMETHASONE SODIUM PHOSPHATE INJECTION USP contains 8 mg/ml of creatinine, which can complicate the evaluation of creatinine clearance.
Pregnancy : Category C
Since adequate human reproduction studies have not been done with corticosteroids, use of these drugs in pregnancy or in women of childbearing potential requires that the anticipated benefits be weighed against the possible hazards to the mother and embryo or foetus. Infants born of mothers who have received substantial doses of corticosteroids during pregnancy should be carefully observed for signs of hypoadrenalism.
Nursing mothers :
Corticosteroids appear in breast milk and could suppress growth, interfere with endogenous corticosteroid production or cause other unwanted effects. Mothers taking pharmacologic doses of corticosteroids should be advised not to nurse.
Paediatric use :
Growth and development of paediatric patients on prolonged corticosteroid therapy should be carefully followed.
INTERACTIONS :
Co-administration of thalidomide with DEXAMETHASONE SODIUM PHOSPHATE INJECTION USP should be employed cautiously, as toxic epidermal necrolysis has been reported with concomitant use. Aspirin should be used cautiously in conjunction with corticosteroids in hypoprothrombinaemia. The renal clearance of salycilates is increased by corticosteroids and therefore salycilate dosage should be reduced along with steroid withdrawal. Cytochrome P450 3A4 (CYP 3A4) enzyme inducers, such as phenytoin, barbiturates (e.g., phenobarbital), carbamazepine, and rifampin may enhance the metabolic clearance of corticosteroids resulting in decreased blood levels and lessened physiologic activity, thus requiring an increase in corticosteroid dosage.
Dexamethasone is metabolized by CYP 3A4. Concomitant administration of dexamethasone with inducers of CYP 3A4 (as listed above) has the potential to result in decreased plasma concentrations of dexamethasone. In addition, concomitant administration of dexamethasone with known inhibitors of CYP 3A4 (e.g., ketoconazole, macrolide antibiotics such as erythromycin) has the potential to result in increased plasma concentrations of dexamethasone. Effects of other drugs on the metabolism of dexamethasone may interfere with dexamethasone suppression tests, which should be interpreted with caution during administration of such drugs. Dexamethasone is a moderate inducer of CYP 3A4. Co-administration of dexamethasone with other drugs that are metabolized by CYP 3A4 (e.g., indinavir, erythromycin) may increase their clearance, resulting in decreased plasma concentrations. In post-marketing experience, there have been reports of both increases and decreases in phenytoin levels with dexamethasone co-administration, leading to alterations in seizure control. Although ketoconazole may increase dexamethasone plasma concentrations through inhibition of CYP 3A4, ketoconazole alone can inhibit adrenal corticosteroid synthesis and may cause adrenal insufficiency during corticosteroid withdrawal.
Ephedrine may enhance the metabolic clearance of corticosteroids, resulting in decreased blood levels and lessened physiologic activity, thus requiring an increase in corticosteroid dosage. False negative results in the dexamethasone suppression test (DST) in patients being treated with indomethacin have been reported. Thus, results of the DST should be interpreted with caution in these patients. The prothrombin time should be checked frequently in patients who are receiving corticosteroids and coumarin anticoagulants at the same time because of reports that corticosteroids have altered the response to these anticoagulants. Studies have shown that the usual effect produced by adding corticosteroids is inhibition of response to coumarins, although there have been some conflicting reports of potentiation not substantiated by studies. When corticosteroids are administered concomitantly with potassium-depleting diuretics, patients should be observed closely for development of hypokalaemia. The desired effects of hypoglycaemic agents (including insulin) are antagonised by corticosteroids. Corticosteroids may affect the nitroblue-tetrazolium test for bacterial infection and produce false negative results.
INCOMPATIBILITIES :
Dexamethasone sodium phosphate is physically incompatible with daunorubicin, doxorubicin, vancomycin, diphenhydramine (with lorazepam and metoclopramide) and metaraminol bitartrate and should not be admixed with solutions containing these drugs. It is also incompatible with doxapram and glycopyrrolate in syringe and with ciprofloxacin, idarubicin and midazolam in Y-site injections (1 : 1 mixture).
SIDE EFFECTS :
The incidence of predictable undesirable effects, including hypothalamic-pituitary-adrenal suppression, correlates with the relative potency of the drug, dosage, timing of administration and the duration of treatment.
Fluid and electrolyte disturbances :
Sodium retention, fluid retention, congestive heart failure in susceptible patients, potassium loss, hypokalaemic alkalosis, hypertension, increased calcium excretion.
Musculoskeletal :
Muscle weakness, steroid myopathy, loss of muscle mass, osteoporosis (especially in post-menopausal females), vertebral compression fractures, aseptic necrosis of femoral and humeral heads, pathological fracture of long bones, tendon rupture and post-injection flare (following intraarticular use).
Gastrointestinal :
Peptic ulcer with possible perforation and haemorrhage, perforation of the small and large bowel, particularly in patients with inflammatory bowel disease, pancreatitis, abdominal distension, ulcerative oesophagitis, dyspepsia, oesophageal candidiasis.
Dermatological :
Impaired wound healing, thin fragile skin, petechiae and ecchymoses, erythema, striae, telangiectasia, acne, increased sweating, possible suppression of skin tests, burning or tingling especially in the perineal area (after intravenous injection), other cutaneous reactions such as allergic dermatitis, urticaria, angioneurotic oedema and hypo-or hyper-pigmentation.
Neurological :
Convulsions, increased intracranial pressure with papilloedema (pseudotumour cerebri) usually after treatment, vertigo, headache, psychic disturbances (e.g. euphoria, psychological dependence, depression, insomnia); cerebral palsy in pre-term infants.
Endocrine :
Menstrual irregularities, amenorrhoea, development of cushingoid state, suppression of growth in children and adolescents, secondary adrenocortical and pituitary unresponsiveness (particularly in times of stress as in trauma, surgery or illness), decreased carbohydrate tolerance, manifestation of latent diabetes mellitus, increased requirements for insulin or oral hypoglycaemic agents in diabetes, hirsutism.
Anti-inflammatory and immunosuppressive effects :
Increased susceptibility and severity of infections with suppression of clinical symptoms and signs; opportunistic infections, recurrence of dormant tuberculosis.
Ophthalmic :
Posterior subcapsular cataracts, increased intraocular pressure, papilloedema, corneal or scleral thinning, exacerbation of ophthalmic viral disease, glaucoma exophthalmos, rare instances of blindness associated with intralesional therapy around the face and head, retinopathy of prematurity.
Metabolic :
Negative nitrogen balance due to protein catabolism, negative calcium balance.
Cardiovascular:
Myocardial rupture following recent myocardial infarction, hypertrophic cardio-myopathy in low birth weight infants.
Other :
Hypersensitivity, including anaphylaxis has been reported, leucocytosis, thromboembolism, weight gain, increased appetite, nausea, malaise, hiccups and sterile abscess.
Withdrawal symptoms and signs :
Too rapid a reduction of corticosteroid dosage following prolonged treatment can lead to acute adrenal insufficiency, hypotension and death. In some instances, withdrawal symptoms may simulate a clinical relapse of the disease for which the patient has been undergoing treatment.
EFFECTS ON ABILITY TO DRIVE AND USE MACHINES :
None reported.
INFORMATION FOR PATIENTS :
Susceptible patients who are on immunosuppressant doses of corticosteroids should be warned to avoid exposure to chickenpox or measles. Patients should also be advised that if they are exposed, medical advice should be sought without delay.
OVERDOSAGE :
Symptoms :
Reports of acute toxicity and/or deaths following overdosage with glucocorticoids are rare. Exaggeration of corticosteroid related adverse effects may occur including hypertension, oedema, peptic ulceration, hyperglycaemia and altered mental state. Anaphylactic or hypersensitivity reactions may occur.
TREATMENT OF OVERDOSAGE :
No antidote is available. Treatment of overdosage is symptomatic. The dosage should be reduced or the drug withdrawn. Anaphylactic and hypersensitivity reactions may be treated with adrenaline (epinephrine), positive pressure artificial respiration and aminophylline. The patient should be kept warm and quiet.
PHARMACEUTICAL PRECAUTIONS :
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit.
STORAGE :
Store below 30°C (86°F), protected from light. Do not freeze. Sensitive to heat. Do not autoclave.
SHELF LIFE :
24 months from the date of manufacture.
PRESENTATION :
DEXAMETHASONE SODIUM PHOSPHATE INJECTION USP contains Dexamethasone Sodium Phosphate USP equivalent to Dexamethasone Phosphate 4 mg in 1 ml aqueous solution.
Such 10 ampoules of 1 ml are packed in a Box.
Disclaimer : For the use of a Registered Medical Practitioner or a Hospital or a Institution only. Also it is not intended to be used by healthcare professionals or patients for the purpose of prescribing or administering these products. Questions regarding the complete and current content of product labeling / specification / presentation should be directed to SGPharma.

 Cardiovascular
Cardiovascular





