
250 mg/5 ml
For the use of a Registered Medical Practitioner or a Hospital or a Institution only.
DIMENHYDRINATE INJECTION USP (Dimenhydrinate) is an antiemetic. Chemically, Dimenhydrinate is 1H -Purine-2,6-dione, 8-chloro-3,7-dihydro-1,3-dimethyl-, compd. with 2-(diphenylmethoxy)-N,N-dimethylethanamine (1:1). The molecular formula is
C17H21NO·C7H7ClN4O2 and molecular weight is 469.96.
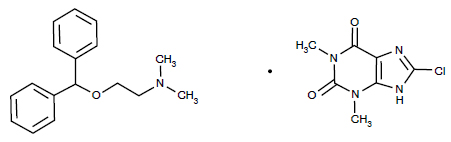
STRUCTURAL FORMULA :
Its structural formula is :
DIMENHYDRINATE INJECTION USP is a sterile, clear, colourless solution filled in amber vial of suitable size.
COMPOSITION :
Each ml contains :
Dimenhydrinate USP 50 mg
Propylene Glycol USP 50 % V/V
Benzyl Alcohol USP 5 % V/V
(as preservative)
Water for Injection USP q.s.
ACTIONS :
Dimenhydrinate, a monoethanolamine derivative, is a sedating antihistamine with antimuscarinic and significant sedative effects. While the precise mode of action of dimenhydrinate is not known, it has a depressant action on hyperstimulated labyrinthine function.
PHARMACOKINETICS :
Distribution :
Small amounts excreted in breast milk.
Elimination :
Renally eliminated.
Onset :
20 to 30 min (Intramuscular).
Duration :
3 to 6 h.
INDICATIONS :
DIMENHYDRINATE INJECTION USP is used mainly as an antiemetic in the prevention and treatment of motion sickness. It is also used for the treatment of nausea and vertigo caused by Meniere’s disease and other vestibular disturbances.
Administration :
DIMENHYDRINATE INJECTION USP is for Intramuscular/ Slow Intravenous Injection.
- Dimenhydrinate in the injectable form is indicated when the oral form is impractical.
- When administering drug I.M., use Z-track method to avoid subcutaneous irritation.
- When administering drug I.V., confirm correct catheter or needle placement. Note that this drug should never be given intra-arterially.
Dosage :
Adults :
Nausea or vomiting may be expected to be controlled for approximately 4 hours with 50 mg, and prevented by a similar dose every 4 hours. Its administration may be attended by some degree of drowsiness in some patients, and 100 mg every 4 hours may be given in conditions in which drowsiness is not objectionable or is even desirable. For intramuscular administration, each milliliter (50 mg) of solution is injected as needed, but for intravenous administration, each milliliter (50 mg) of solution must be diluted in 10 ml of 0.9 % Sodium Chloride Injection, USP and injected over a period of 2 minutes.
Paediatric :
For intramuscular administration, 1.25 mg/kg of body weight or 37.5 mg/m2 of body surface area is administered four times daily. The maximum dose should not exceed 300 mg daily.
CONTRAINDICATIONS :
Neonates and patients with a history of hypersensitivity to dimenhydrinate or its components (diphenhydramine or 8-chlorotheophylline) should not be treated with dimenhydrinate.
WARNINGS :
Caution should be used when dimenhydrinate is given in conjunction with certain antibiotics that may cause ototoxicity, since dimenhydrinate is capable of masking ototoxic symptoms, and an irreversible state may be reached. This drug may impair the mental and/or physical abilities required for the performance of potentially hazardous tasks, such as driving a vehicle or operating machinery. The concomitant use of alcohol or other central nervous system depressants may have an additive effect. Therefore, patients should be warned accordingly. Dimenhydrinate should be used with caution in patients having conditions which might be aggravated by anticholinergic therapy (i.e., prostatic hypertrophy, stenosing peptic ulcer, pyloroduodenal obstruction, bladder neck obstruction, narrow-angle glaucoma, bronchial asthma, or cardiac arrhythmias). The preparation should not be injected intra-arterially. DIMENHYDRINATE INJECTION USP contains benzyl alcohol as preservative. Benzyl alcohol has been reported to be associated with a fatal “Gasping Syndrome” in premature infants. Symptoms include a striking onset of gasping syndrome, hypotension, bradycardia and cardiovascular collapse.
Paediatric Patients :
For infants and children especially, antihistamines in overdosage may cause hallucinations, convulsions, or death. As in adults, antihistamines may diminish mental alertness in paediatric patients. In the young child, particularly, they may produce excitation.
PRECAUTIONS :
General :
Drowsiness may be experienced by some patients, especially with high dosage. This effect frequently is not undesirable in conditions for which the drug is used.
Carcinogenesis, Mutagenesis, Impairment Of Fertility :
Mutagenicity screening tests performed with dimenhydrinate, diphenhydramine, and 8-chlorotheophylline produced positive results in the bacterial systems and negative results in the mammalian systems. There are no human data that indicate dimenhydrinate is a carcinogen or mutagen or that it impairs fertility.
Pregnancy : Category B
Reproduction studies have been performed in rats at doses up to 20 times the human dose, and in rabbits at doses up to 25 times the human dose (on a mg/kg basis), and have revealed no evidence of impaired fertility or harm to the foetus due to dimenhydrinate. There are no adequate and well-controlled studies in pregnant women. However, clinical studies in pregnant women have not indicated that dimenhydrinate increases the risk of abnormalities when administered in any trimester of pregnancy. It would appear that the possibility of foetal harm is remote when the drug is used during pregnancy. Nevertheless, because the studies in humans cannot rule out the possibility of harm, dimenhydrinate should be used during pregnancy only if clearly needed.
Nursing mothers :
Small amounts of dimenhydrinate are excreted in breast milk. Because of the potential for adverse reactions in nursing infants from dimenhydrinate, a decision should be made whether to discontinue nursing or to discontinue the drug, taking into account the importance of the drug to the mother.
Paediatric Use :
Safety and efficacy in children younger than 2 years of age not established.
INTERACTIONS :
Alcohol, CNS depressants :
Enhances CNS depressant effects.
Aminoglycosides :
May mask signs of aminoglycoside-related ototoxicity.
Anticholinergic drugs :
Causes additive anticholinergic effects.
Laboratory Test Interactions :
May cause false elevation in serum theophylline levels.
INCOMPATIBILITY :
Ammonium chloride, amobarbital, butorphanol, chlorpromazine, glycopyrrolate, heparin, hydrocortisone, hydroxyzine, midazolam, pentobarbital, phenobarbital, phenytoin, prednisolone, prochlorperazine, promethazine, tetracycline, theophylline, thiopental, trifluoperazine.
SIDE EFFECTS :
Cardiovascular :
Palpitations, hypotension, tachycardia.
Central Nervous System :
Sedation, hallucinations, delirium, drowsiness, confusion, nervousness, restlessness, headache, insomnia, tingling, heaviness and weakness of hands, vertigo, dizziness, lassitude, excitation.
Dermatologic :
Fixed drug eruption, photosensitivity.
Eye, Ear, Nose and Throat :
Diminished night vision, decreased colour discrimination, exacerbation of narrow-angle glaucoma, blurred vision, diplopia, nasal stuffiness, dryness of nose and throat.
Gastro-intestinal :
Nausea, vomiting, diarrhoea, GI distress, constipation, anorexia, dry mouth.
Genitourinary :
Prostatic enlargement, difficult or painful urination.
Respiratory :
Tightness of chest, wheezing, thickening of bronchial secretions.
Other:
Anaphylaxis.
EFFECTS ON ABILITY TO DRIVE AND USE MACHINES :
Because of the potential for drowsiness, patients taking dimenhydrinate should be cautioned against operating automobiles or dangerous machinery.
OVERDOSAGE :
Drowsiness is the usual clinical side effect. Convulsions, coma, and respiratory depression may occur with massive overdosage.
TREATMENT OF OVERDOSAGE :
No specific antidote is known. If respiratory depression occurs, mechanically assisted respiration should be initiated and oxygen should be administered. Convulsions should be treated with appropriate doses of diazepam. Phenobarbital (5 to 6 mg/kg) may be given to control convulsions in paediatric patients.
PHARMACEUTICAL PRECAUTIONS :
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit.
STORAGE :
Store below 30°C (86°F), protected from light. Do not refrigerate.
SHELF LIFE :
24 months from the date of manufacture.
PRESENTATION :
DIMENHYDRINATE INJECTION USP is supplied as 250 mg Dimenhydrinate USP in 5 ml aqueous solution.
Such 1 vial of 5 ml is packed in a Box.
Disclaimer : For the use of a Registered Medical Practitioner or a Hospital or a Institution only. Also it is not intended to be used by healthcare professionals or patients for the purpose of prescribing or administering these products. Questions regarding the complete and current content of product labeling / specification / presentation should be directed to SGPharma.

 Cardiovascular
Cardiovascular





