
7.5 mg
For the use of a Registered Medical Practitioner or a Hospital or a Institution only.
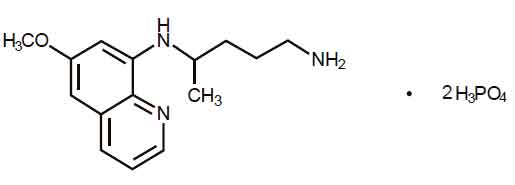
PRIMAQUINE PHOSPHATE TABLETS USP (Primaquine Phosphate) is a synthetic compound with potent antimalarial activity. Chemically, Primaquine Phosphate is(±)-8-[(4-Amino-1-methylbutyl)amino]-6-methoxyquinoline phosphate (1:2). The molecular formula is C15H21N3O · 2H3PO4 and molecular weight is 455.34.
STRUCTURAL FORMULA :
Its structural formula is :
PRIMAQUINE PHOSPHATE TABLETS USP are pink coloured, circular, biconvex film coated tablet having ‘SGP’ embossed on one side and breakline on other side.
COMPOSITION :
Each film coated tablet contains :
Primaquine Phosphate USP 13.2 mg
equivalent to Primaquine 7.5 mg
Excipients q.s.
Colours : Red Ferric Oxide NF, Titanium Dioxide USP
ACTIONS :
Primaquine phosphate is an 8-amino-quinoline compound which eliminates tissue (exoerythrocytic) infection. Thereby, it prevents the development of the blood (erythrocytic) forms of the parasite which are responsible for relapses in vivax malaria. Primaquine phosphate is also active against gametocytes of Plasmodium falciparum.
PHARMACOKINETICS :
Primaquine is readily absorbed from the gastrointestinal tract. Peak plasma concentrations occur about 1 to 2 hours after a dose is taken and then rapidly diminish with a reported elimination half-life of 3 to 6 hours. It is widely distributed into body tissues. Primaquine is rapidly metabolised in the liver, its major metabolite being carboxyprimaquine, and little unchanged drug is excreted in the urine. Carboxyprimaquine accumulates in the plasma on repeated dosage.
INDICATIONS :
PRIMAQUINE PHOSPHATE TABLETS USP is indicated for adjunct in the treatment of Plasmodium vivax and P. ovale malaria (eradication of liver stages).
Administration :
PRIMAQUINE PHOSPHATE TABLETS USP is for oral administration. Taking PRIMAQUINE PHOSPHATE TABLETS USP after a meal may reduce abdominal pain or cramps associated with ingestion of the drug.
Dosage :
Adult :
P. vivax infection primaquine is given in a dose of 30 mg daily for 14 days and for P. ovale infection it is given in a dose of 15 mg daily for 14 days.
Children :
For a radical cure, primaquine is then given to children over 6 months of age; specialist advice should be sought for children under 6 months of age. In P. vivax infection primaquine is given in a dose of 500 micrograms/kg (maximum 30 mg) daily for 14 days, and for P. ovale infection it is given in a dose of 250 micrograms/kg (maximum 15 mg) daily for 14 days. Before starting primaquine, blood should be tested for glucose-6-phosphate dehydrogenase (G6PD) activity since the drug can cause haemolysis in G6PD-deficient patients. Specialist advice should be obtained in G6PD deficiency; in mild G6PD deficiency primaquine in a dose for adults of 45 mg once a week (children 750 micrograms/kg once a week; maximum 45 mg once a week) for 8 weeks, has been found useful and without undue harmful effects.
CONTRAINDICATIONS :
PRIMAQUINE PHOSPHATE TABLETS USP is contraindicated :
In patients who are hypersensitive to primaquine or to any ingredient in the formulation or components of the container. In acutely ill patients suffering from systemic disease manifested by tendency to granulocytopenia, such as rheumatoid arthritis and lupus erythematosus.
In patients receiving concurrently other potentially haemolytic drugs or depressants of myeloid elements of the bone marrow. Quinacrine appears to potentiate the toxicity of antimalarial compounds which are structurally related to primaquine; therefore, the use of quinacrine in patients receiving primaquine is contraindicated. Similarly, PRIMAQUINE PHOSPHATE TABLETS USP should not be administered to patients who have received quinacrine recently, as toxicity is increased.
Discontinue the use of PRIMAQUINE PHOSPHATE TABLETS USP promptly if signs suggestive of haemolytic anaemia occur (darkening of the urine, marked fall of haemoglobin or erythrocytic count). Haemolytic reactions (moderate to severe) may occur in individuals with glucose-6-phosphate dehydrogenase (G-6-PD) deficiency and in individuals with a family or personal history of favism. Areas of high prevalence of G-6-PD deficiency are Africa, Southern Europe, Mediterranean region, Middle East, South-East Asia, and Oceania. People from these regions have a greater tendency to develop haemolytic anaemia (due to a congenital deficiency of erythrocytic glucose-6-phosphate dehydrogenase) while receiving Primaquine and related drugs.
PRECAUTIONS :
Since anaemia, methaemoglobinaemia, and leucopenia have been observed following administration of large doses of primaquine, the adult dosage of 15 mg daily for fourteen days should not be exceeded. It is also advisable to make routine blood examinations (particularly blood cell counts and haemoglobin determinations) during therapy.
If PRIMAQUINE PHOSPHATE TABLETS USP is prescribed for :
1. An individual who has shown a previous idiosyncrasy to primaquine phosphate (as manifested by haemolytic anaemia, methaemoglobinaemia, or leucopenia),
2. an individual with a family or personal history of favism, or
3. an individual with erythrocytic glucose-6-phosphate dehydrogenase (G-6-PD) deficiency or nicotinamide adenine dinucleotide (NADH) methaemoglobin reductase deficiency, the person should be observed closely for tolerance.
The drug should be discontinued immediately if marked darkening of the urine or sudden decrease in haemoglobin concentration or leucocyte count occurs.
Pregnancy :
Safe usage of this preparation in pregnancy has not been established. Therefore, use of it during pregnancy should be avoided except when in the judgment of the physician the benefit outweighs the possible hazard.
Nursing mothers :
It is not known whether primaquine is excreted in breast milk. Because of the potential of primaquine to produce serious adverse reactions in nursing infants, a decision should be made whether to discontinue breast-feeding or to discontinue the drug.
Paediatric Use :
Children should avoid travel to areas with chloroquine-resistant P. falciparum unless they can take a highly effective antimalarial agent, such as primaquine. Primaquine is safe and highly effective in children provided that the glucose-6-phosphate dehydrogenase
(G-6-PD) level is normal.
INTERACTIONS AND INCOMPATIBILITIES :
Primaquine should not be used with drugs liable to induce haemolysis or bone marrow depression. Theoretically, mepacrine may increase the plasma concentrations of primaquine resulting in a higher risk of toxicity, and it has been recommended that these drugs should not be used together. Primaquine inhibits hepatic drug oxidation and is reported to inhibit the metabolism of loroquine. Quinacrine may potentiate toxicity of antimalarial compounds that are structurally related to primaquine. Caution is advised if primaquine is used concomitantly with other drugs that prolong the QT interval.
SIDE EFFECTS :
Gastro-intestinal :
Abdominal pain and gastric distress are more common if taken on an empty stomach. Larger doses may cause nausea and vomiting.
Haematologic :
Methaemoglobinaemia may occur occasionally. Haemolytic anaemia can occur in persons with glucose-6-phosphate dehydrogenase (G-6-PD) deficiency. Other uncommon effects include mild anaemia and leucocytosis. Primaquine may rarely produce leucopenia or agranulocytosis, usually after overdosage.
Cardiovascular :
Hypertension and cardiac arrhythmias have been reported on rare occasions.
EFFECTS ON ABILITY TO DRIVE AND USE
MACHINES :
Not known.
OVERDOSAGE :
Symptoms :
Abdominal cramps, vomiting, jaundice, burning epigastric distress, CNS and cardiovascular disturbances, including cardiac arrhythmia and QT prolongation, cyanosis, methaemoglobinaemia, moderate leucocytosis or leucopenia, and anaemia. The most striking changes are granulocytopenia and acute haemolytic anaemia in sensitive persons. Acute haemolysis often occurs, but complete recovery can be expected if primaquine is discontinued.
TREATMENT OF OVERDOSAGE :
Management should include appropriate attempts to recover primaquine from the stomach by emesis or gastric lavage and provision of respiratory and cardiovascular support. Sodium lactate intravenous may be used to counter the depressant effects of primaquine on the heart. Electrical pacing of the heart may be needed. Ammonium chloride in doses up to 12 g daily orally may be given to enhance urinary excretion. Symptomatic methaemoglobinaemia should be treated with 1 to 2 mg per kg of methylene blue.
STORAGE :
Store below 30°C (86°F), protected from moisture and light.
Do not refrigerate.
SHELF LIFE :
24 months from the date of manufacture.
PRESENTATION :
PRIMAQUINE PHOSPHATE TABLETS USP contains Primaquine Phosphate USP 13.2 mg equivalent to Primaquine 7.5 mg.
1 Strip of 10 Tablets per Box.
Disclaimer : For the use of a Registered Medical Practitioner or a Hospital or a Institution only. Also it is not intended to be used by healthcare professionals or patients for the purpose of prescribing or administering these products. Questions regarding the complete and current content of product labeling / specification / presentation should be directed to SGPharma.

 Cardiovascular
Cardiovascular





