5,000 IU/5 ml, 25,000 IU/5 ml
For the use of a Registered Medical Practitioner or a Hospital or a Institution only.
BLOCKASOL (Heparin) is a heterogenous group of straight-chain anionic mucopolysaccharides, called glycosaminoglycans having anticoagulant properties. Although others may be present, the main sugars occurring in heparin are : (1) α-L-iduronic acid 2-sulfate, (2) 2-deoxy-2-sulfamino-α-D-glucose 6-sulfate, (3) β-D-glucuronic acid, (4) 2-acetamido-2-deoxy-α-D-glucose, and (5) α-L-iduronic acid. These sugars are present in decreasing amounts, usually in the order (2) > (1) > (4) > (3) > (5), and are joined by glycosidic linkages, forming polymers of varying sizes.
STRUCTURAL FORMULA :
Its structural formula is :
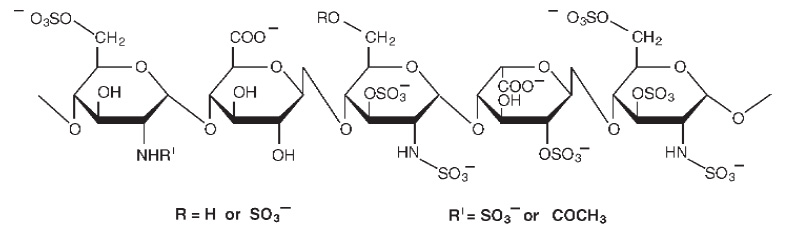
BLOCKASOL is sterile solution of heparin sodium derived from porcine mucosa, standardized for anticoagulant activity.
COMPOSITION :
Each ml contains :
Heparin Sodium I.P
equivalent to Heparin
units (from Mucosa) 1,000 IU
Benzyl Alcohol I.P. 0.95 % v/v
Water for Injections I.P. q.s.
Each ml contains :
Heparin Sodium I.P.
equivalent to Heparin
units (from Mucosa) 5,000 IU
Benzyl Alcohol I.P. 0.95 % v/v
Water for Injections I.P. q.s.
ACTIONS :
Heparin inhibits reactions that lead to the clotting of blood and the formation of fibrin clots both in vitro and in vivo. Heparin acts at multiple sites in the normal coagulation system. Small amounts of heparin in combination with antithrombin III (heparin cofactor) can inhibit thrombosis by inactivating activated Factor X and inhibiting the conversion of prothrombin to thrombin. Once active thrombosis has developed, larger amounts of heparin can inhibit further coagulation by inactivating thrombin and preventing the conversion of fibrinogen to fibrin. Heparin also prevents the formation of a stable fibrin clot by inhibiting the activation of the fibrin stabilizing factor.
PHARMACOKINETICS :
Heparin is not absorbed from the G.I. tract. Onset of anti coagulation is immediate with I.V. use and within 20 - 30 minutes with subcutaneous use. Peak plasma levels of Heparin Sodium are achieved 2 to 4 hours following subcutaneous administration, although considerable individual variations are possible. Biotransformation takes place in the liver and the reticulo-endothelial system. Heparin Sodium does not have fibrinolytic activity; therefore, it will not lyse the existing clots. Heparin Sodium does not cross the placental barrier and is not distributed in human milk. Mean half-life is 1.5 hours. Elimination is by renal excretion mainly as metabolites with small amounts being excreted unchanged in urine.
INDICATIONS :
BLOCKASOL is indicated for :
Anticoagulant therapy in prophylaxis and treatment of venous thrombosis and its extension;
(In a low-dose regimen) for prevention of postoperative deep venous thrombosis and pulmonary embolism in patients undergoing major abdomino-thoracic surgery or who for other reasons are at risk of developing thromboembolic disease;
Prophylaxis and treatment of pulmonary embolism;
Atrial fibrillation with embolization;
Diagnosis and treatment of acute and chronic consumption coagulopathies (disseminated intravascular coagulation);
Prevention of clotting in arterial and heart surgery;
Prophylaxis and treatment of peripheral arterial embolism;
As an anticoagulant in blood transfusions, extracorporeal circulation and dialysis procedures and in blood samples for laboratory purposes.
When heparin is added to an infusion solution for continuous intravenous administration, the container should be inverted at least 6 times to ensure adequate mixing and prevent pooling of the heparin in the solution.
Administration :
Heparin sodium is not effective by oral administration and should be given by intermittent intravenous injection, intravenous infusion, or deep subcutaneous (intrafat i.e. above the iliac crest or abdominal fat layer) injection. The intramuscular route of administration should be avoided because of the frequent occurrence of haematoma at the injection site.
Dosage :
The dosage of BLOCKASOL should be adjusted according to the patient’s coagulation test results. When BLOCKASOL is given by continuous intravenous infusion, the coagulation time should be determined approximately every 4 hours in the early stages of treatment. When the drug is administered intermittently by intravenous injection, coagulation tests should be performed before each injection during the early stages of treatment and at appropriate intervals thereafter. Dosage is considered adequate when the activated partial thromboplastin time (APTT) is 1.5 to 2 times normal or when the whole blood clotting time is elevated approximately 2.5 to 3 times the control value. After deep subcutaneous (intrafat) injections, tests for adequacy of dosage are best performed on samples drawn 4 - 6 hours after the injections. Periodic platelet counts, haematocrits, and tests for occult blood in stool are recommended during the entire course of heparin therapy, regardless of the route of administration. BLOCKASOL should not be mixed with doxorubicin, droperidol, ciprofloxacin, or mitoxantrone, since it has been reported that these drugs are incompatible with heparin and a precipitate may form.
Converting to Oral Anticoagulant :
When an oral anticoagulant of the coumarin or similar type is to be begun in patients already receiving heparin sodium, baseline and subsequent tests of prothrombin activity must be determined at a time when heparin activity is too low to affect the prothrombin time. This is about 5 hours after the last I.V. bolus and 24 hours after the last subcutaneous dose. If continuous I.V. heparin infusion is used, prothrombin time can usually be measured at any time. In converting from heparin to an oral anticoagulant, the dose of the oral anticoagulant should be the usual initial amount and thereafter prothrombin time should be determined at the usual intervals. To ensure continuous anticoagulation, it is advisable to continue full heparin therapy for several days after the prothrombin time has reached the therapeutic range. Heparin therapy may then be discontinued without tapering.
Therapeutic Anticoagulant Effect with Full-Dose Heparin : Although dosage must be adjusted for the individual patient according to the results of suitable laboratory tests, the following dosage schedules may be used as guidelines :
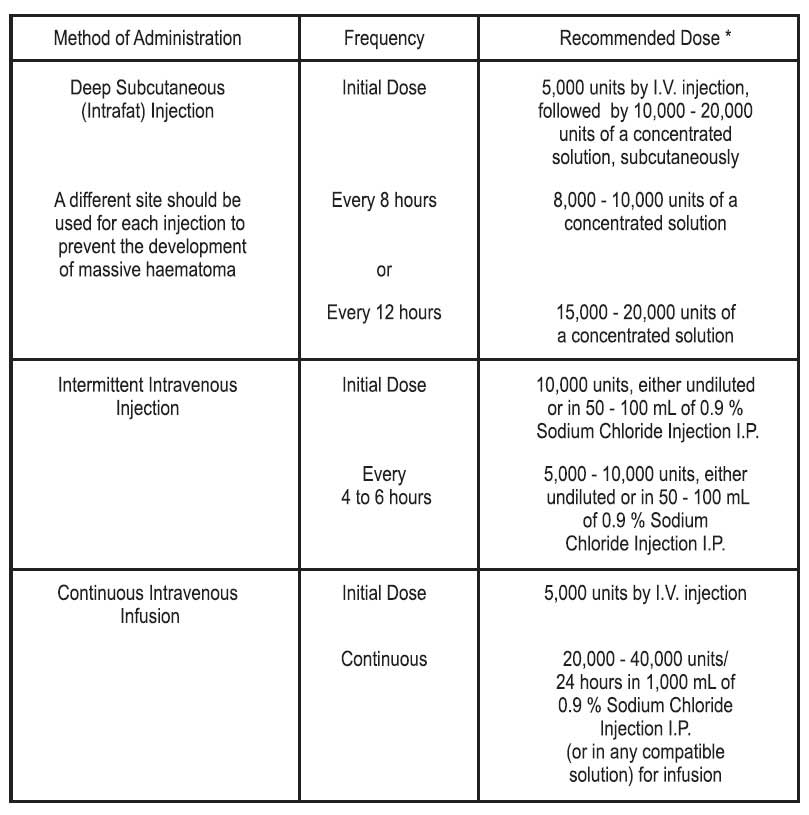
* Based on 150-lb. (68-kg) patient.
Paediatric Use :
Follow recommendations of appropriate paediatric reference texts. In general, the following dosage schedule may be used as a guideline :
Initial Dose : 50 units/kg (I.V., drip)
Maintenance Dose : 100 units/kg (I.V., drip) every 4 hours or
20,000 units/M2/24 hours continuously.
Surgery of the Heart and Blood Vessels :
Patients undergoing total body perfusion for open-heart surgery should receive an initial dose of not less than 150 units of heparin sodium per kg of body weight. Frequently, a dose of 300 units per kg is used for procedures estimated to last less than 60 minutes or 400 units per kg for those estimated to last longer than 60 minutes.
CONTRAINDICATIONS :
Heparin should not be used in-patients with hypersensitivity to heparin or any component; severe thrombocytopenia, subacute bacterial endocarditis, suspected intracranial haemorrhage, uncontrollable bleeding (unless secondary to disseminated intravascular coagulation). Also Heparin should not be used in patients in whom suitable blood coagulation tests e.g. the whole-blood clotting time, partial thromboplastin time, etc. cannot be performed at appropriate intervals.
WARNINGS :
Heparin is not intended for intramuscular use.
Hypersensitivity :
Patients with documented hypersensitivity to heparin should be given the drug only in clearly life-threatening situations. Haemorrhage : Haemorrhage can occur at virtually any site in patients receiving heparin. An unexplained fall in haematocrit, fall in blood pressure, or any other unexplained symptom should lead to serious consideration of a haemorrhagic event. Heparin sodium should be used with extreme caution in disease states in which there is increased danger of haemorrhage. Some of the conditions in which increased danger of haemorrhage exists are :
Coagulation Testing :
When heparin sodium is administered in therapeutic amounts, its dosage should be regulated by frequent blood coagulation tests. If the coagulation test is unduly prolonged or if haemorrhage occurs, heparin sodium should be discontinued promptly.
Thrombocytopenia :
Thrombocytopenia has been reported to occur in patients receiving heparin with a reported incidence of 0 to 30 %. Mild thrombocytopenia (count greater than 100,000/mm3) may remain stable or reverse even if heparin is continued. However, reduction in platelet count of any degree should be monitored closely. If the count falls below 100,000/mm3 or if recurrent thrombosis develops, the heparin product should be discontinued. If continued heparin therapy is essential, administration of heparin from a different organ source can be reinstituted with caution.
This product contains benzyl alcohol as preservative. Benzyl alcohol has been reported to be associated with a fatal “Gasping Syndrome” in premature infants. Symptoms include a striking onset of gasping syndrome, hypotension, bradycardia and cardiovascular collapse.
Usage in Pregnancy :
Teratogenic Effects : Pregnancy Category C. Animal reproduction studies have not been conducted with heparin sodium. It is also not known whether heparin sodium can cause foetal harm when administered to a pregnant woman or can affect reproduction capacity. Heparin sodium should be given to a pregnant woman only if clearly needed. Nonteratogenic Effects : Heparin does not cross the placental barrier.
Nursing Mothers :
Heparin is not excreted in human milk.
INTERACTION :
a. Drugs Enhancing Heparin Effect :
Oral anticoagulants : Heparin sodium may prolong the one-stage prothrombin time. Therefore, when heparin sodium is given with dicumarol or warfarin sodium, a period of at least 5 hours after the last intravenous dose or 24 hours after the last subcutaneous dose should elapse before blood is drawn if a valid prothrombin time is to be obtained. Platelet inhibitors : Drugs such as acetylsalicylic acid, dextran, phenylbutazone, ibuprofen, indomethacin, dipyridamole, hydroxychloroquine and others that interfere with platelet aggregation reactions (the main haemostatic defense of heparinized patients) may induce bleeding and should be used with caution in patients receiving heparin sodium. The anticoagulant effect of heparin is enhanced by concurrent treatment with antithrombin III (human) in patients with hereditary antithrombin III deficiency. Thus in order to avoid bleeding, reduced dosage of heparin is recommended during treatment with antithrombin III (human).
b. Drugs Decreasing Heparin Effect :
Digitalis, tetracyclines, nicotine, or antihistamines may partially counteract the anticoagulant action of heparin sodium. Heparin Sodium Injection should not be mixed with doxorubicin, droperidol, ciprofloxacin, or mitoxantrone, since it has been reported that these drugs are incompatible with heparin and a precipitate may form.
SIDE EFFECTS :
1. Haemorrhage : Haemorrhage is the chief complication that may result from heparin sodium therapy, withdrawing the drug can usually control an overly prolonged clotting time or minor bleeding during therapy. Adrenal haemorrhage, Ovarian (corpus luteum) haemorrhage and Retroperitoneal haemorrhage are also known to occur.
2. Local irritation : Local irritation, erythema, mild pain, haematoma or ulceration may follow deep subcutaneous (intrafat) injection of heparin sodium. These complications are much more common after intramuscular use, and such use is not recommended.
3. Hypersensitivity : Generalized hypersensitivity reactions have been reported, with chills, fever, and urticaria as the most usual manifestations and asthma, rhinitis, lacrimation, headache, nausea and vomiting, and anaphylactoid reactions, including shock, occurring more rarely. Itching and burning, especially on the plantar site of the feet, may occur. Thrombocytopenia has been reported to occur in patients receiving heparin with a reported incidence of 0 - 30 %. While often mild and of no obvious clinical significance, a reduction in platelet count can be accompanied by severe thromboembolic complications such as skin necrosis, gangrene of the extremities that may lead to amputation, myocardial infarction, pulmonary embolism, stroke, and possibly death. Certain episodes of painful, ischemic, and cyanosed limbs have in the past been attributed to allergic vasospastic reactions. Whether these are in fact identical to the thrombocytopenia associated complications remains to be determined.
4. Miscellaneous : Osteoporosis following long-term administration of high doses of heparin, cutaneous necrosis after systemic administration, suppression of aldosterone synthesis, delayed transient alopecia, priapism and rebound hyperlipemia on discontinuation of heparin sodium have also been reported. Significant elevations of aminotransferase (SGOT [S-AST] and SGPT [S-ALT]) levels have occurred in a high percentage of patients (and healthy subjects) who have received heparin.
OVERDOSAGE AND TREATMENT OF OVERDOSAGE :
Symptoms : Bleeding is the chief sign of heparin overdosage. Nosebleeds, blood in urine or tarry stools may be noted as the first sign of bleeding. Easy bruising or petechial formations may precede frank bleeding.
Treatment : Neutralization of heparin effect. When clinical circumstances (bleeding) require reversal of heparinization, protamine sulfate (1 % solution) by slow infusion will neutralize heparin sodium. No more than 50 mg should be administered, very slowly, in any 10 minute period. Each mg of protamine sulfate neutralizes approximately 100 I.P. heparin units. The amount of protamine required decreases over time as heparin is metabolized. Although the metabolism of heparin is complex, it may, for the purpose of choosing a protamine dose, be assumed to have a half life of about 1/2 hour after intravenous injection. Administration of protamine sulfate can cause severe hypotensive and anaphylactoid reactions. Because fatal reactions often resembling anaphylaxis have been reported, the drug should be given only when resuscitation techniques and treatment of anaphylactoid shock are readily available.
PHARMACEUTICAL PRECAUTIONS :
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit. Slight discoloration does not alter potency.
STORAGE :
Store in a cool place.
Do not freeze.
SHELF LIFE :
24 months from the date of manufacture.
PRESENTATION :
BLOCKASOL, Heparin Sodium Injection I.P. derived from porcine mucosa is available in the following strengths and pack sizes :
1,000 units per ml
5 ml vials
10 ml vials
5,000 units per ml
5 ml vials

 Cardiovascular
Cardiovascular