
200 mg
For the use of a Registered Medical Practitioner or a Hospital or a Institution only.
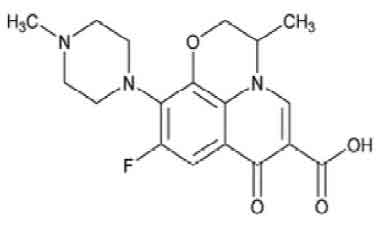
OFLOXACIN TABLETS USP is a synthetic broad-spectrum antimicrobial agent. Chemically, Ofloxacin is (±)-9-Fluoro-2,3-dihydro-3-methyl-10-(4-methyl-1piperazinyl)-7-oxo-7H-pyrido [1,2,3-de]-1,4-benzoxazine-6-carboxylic acid. The molecular formula is C18H20FN3O4 and molecular weight is 361.37.
STRUCTURAL FORMULA :
Its structural formula is :
OFLOXACIN TABLETS USP is white coloured, circular, biconvex film coated tablet.
COMPOSITION :
Each film coated tablet contains :
Ofloxacin USP 200 mg
Excipients q.s.
Colours : Titanium Dioxide B.P., Iron Oxide Yellow.
ACTIONS :
Ofloxacin is a quinolone-carboxylic acid derivative with a wide range of antibacterial activity against both gram negative and gram positive organisms. It is active after oral administration. It inhibits bacterial DNA replication by blocking DNA topo-isomerases, in particular DNA gyrase. Therapeutic doses of ofloxacin are devoid of pharmacological effects on the voluntary or autonomic nervous systems. Microbiological results indicate that the following pathogens may be regarded as sensitive : Staphylococcus aureus (including methicillin resistant staphylococci), Staphylococcus epidermidis, Neisseria species, Escherichia coli, Citrobacter, Klebsiella, Enterobacter, Hafnia, Proteus (indole-negative and indole-positive strains), Haemophilus influenzae, Chlamydiae, Legionella, Gardnerella. Variable sensitivity is shown by Streptococci, Serratia marcescens, Pseudomonas aeruginosa and Mycoplasmas. Anaerobic bacteria (e.g. Fusobacterium species, Bacteroides species, Eubacterium species, Peptococci, Peptostreptococci) are normally resistant.
PHARMACOKINETICS :
Ofloxacin is rapidly and well absorbed from the gastrointestinal tract. Oral bioavailability is almost 100 % and a peak plasma concentration of 3 to 5 micrograms/ml is achieved 1 to 2 hours after a dose of 400 mg by mouth. Absorption may be delayed by the presence of food, but the extent of absorption is not substantially affected. The plasma half-life ranges from 4 to 7 hours; in renal impairment values of 15 to 60 hours have been reported. About 25 % is bound to plasma proteins. Ofloxacin is widely distributed in body fluids, including the CSF, and tissue penetration is good. It crosses the placenta and is distributed into breast milk. It also appears in the bile. There is limited metabolism to desmethyl and N-oxide metabolites; desmethylofloxacin has moderate antibacterial activity. Ofloxacin is eliminated mainly by the kidneys. Excretion is by tubular secretion and glomerular filtration and 65 to 80 % of a dose is excreted unchanged in the urine over 24 to 48 hours, resulting in high urinary concentrations. Less than 5 % is excreted in the urine as metabolites. From 4 to 8 % of a dose may be excreted in the faeces. Only small amounts of Ofloxacin are removed by haemodialysis.
MICROBIOLOGY :
Ofloxacin is a quinolone antimicrobial agent. The mechanism of action of ofloxacin and other fluoroquinolone antimicrobials involves inhibition of bacterial topoisomerase IV and DNA gyrase (both of which are type II topoisomerases), enzymes required for DNA replication, transcription, repair and recombination. Ofloxacin has in vitro activity against a wide range of gram-negative and gram-positive microorganisms. Ofloxacin is often bactericidal at concentrations equal to or slightly greater than inhibitory concentrations.
Fluoroquinolones, including ofloxacin, differ in chemical structure and mode of action from aminoglycosides, macrolides and β-lactam antibiotics, including penicillins. Fluoroquinolones may, therefore, be active against bacteria resistant to these antimicrobials.
Resistance to ofloxacin due to spontaneous mutation in vitro is a rare occurrence (range : 10-9 to 10-11). Although cross-resistance has been observed between ofloxacin and some other fluoroquinolones, some microorganisms resistant to other fluoroquinolones may be susceptible to ofloxacin. Ofloxacin has been shown to be active against most strains of the following microorganisms both in vitro and in clinical infections.
Dilution techniques :
Quantitative methods are used to determine antimicrobial minimum inhibitory concentrations (MIC values). These MIC values provide estimates of the susceptibility of bacteria to antimicrobial compounds. The MIC values should be determined using a standardized procedure. Standardized procedures are based on a dilution method1 (broth or agar) or equivalent with standardized inoculum concentrations and standardized concentrations of ofloxacin powder. The MIC values should be interpreted according to the following criteria :
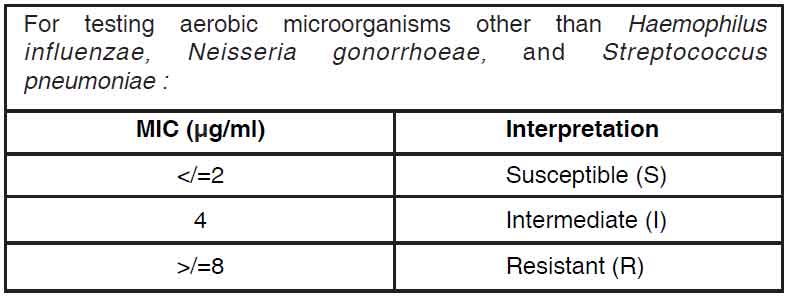
The current absence of data on resistant strains precludes defining any results other than “Susceptible”. Strains yielding MIC results suggestive of a “nonsusceptible” category should be submitted to a reference laboratory for further testing.
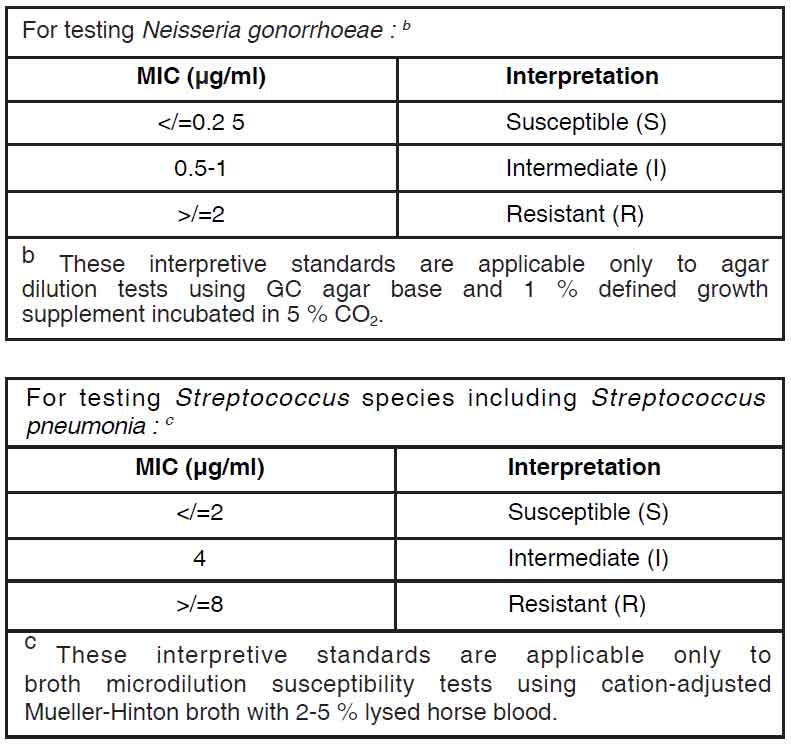
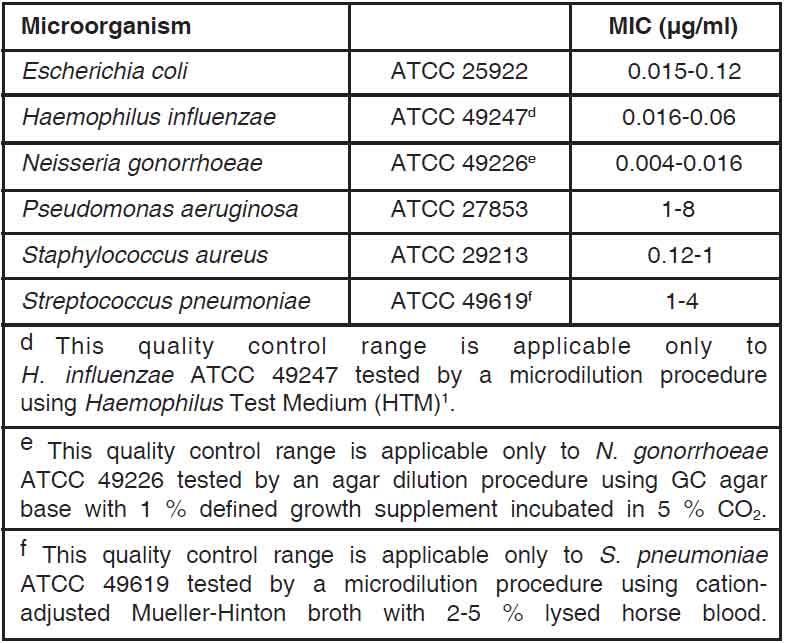
A report of “Susceptible” indicates that the pathogen is likely to be inhibited if the antimicrobial compound in the blood reaches the concentrations usually achievable. A report of “Intermediate” indicates that the result should be considered equivocal, and, if the microorganism is not fully susceptible to alternative, clinically feasible drugs, the test should be repeated. This category implies possible clinical applicability in body sites where the drug is physiologically concentrated or in situations where high dosage of drug can be used. This category also provides a buffer zone which prevents small uncontrolled technical factors from causing major discrepancies in interpretation. A report of “Resistant” indicates that the pathogen is not likely to be inhibited if the antimicrobial compound in the blood reaches the concentrations usually achievable; other therapy should be selected. Standardized susceptibility test procedures require the use of laboratory control microorganisms to control the technical aspects of the laboratory procedures. Standard ofloxacin powder should provide the following MIC values :
Diffusion techniques :
Quantitative methods that require measurement of zone diameters also provide reproducible estimates of the susceptibility of bacteria to antimicrobial compounds. One such standardized procedure 2 requires the use of standardized inoculums concentrations. This procedure uses paper disks impregnated with 5-μg ofloxacin to test the susceptibility of microorganisms to ofloxacin.Reports from the laboratory providing results of the standard single-disk susceptibility test with a 5-μg ofloxacin disk should be interpreted according to the following criteria :
INDICATIONS :
OFLOXACIN TABLETs USP is indicated for the treatment of the following infections when caused by sensitive organisms : Upper and lower urinary tract infections, chronic prostatitis, lower respiratory tract infections, skin and soft tissue infections, uncomplicated
urethral and cervical gonorrhoea, uncomplicated genital chlamydial infection, non-gonococcal urethritis, pelvic inflammatory disease.
Administration : OFLOXACIN TABLETS USP is for oral administration.
Dosage :
Urinary-tract infections : 200-400 mg daily preferably in the morning, increased if necessary in upper urinary-tract infections to 400 mg twice daily.
Chronic prostatitis : 200 mg twice daily for 28 days.
Lower respiratory-tract infections : 400 mg daily preferably in the morning, increased if necessary to 400 mg twice daily.
Skin and soft-tissue infections : 400 mg twice daily.
Uncomplicated gonorrhoea : 400 mg as a single dose.
Uncomplicated genital chlamydial infection, non-gonococcal urethritis : 400 mg daily in single or divided doses for 7 days.
Pelvic inflammatory disease : 400 mg twice daily for 14 days.
Impaired renal function :
Following a normal initial dose, dosage should be reduced in patients with impairment of renal function. When creatinine clearance is 20-50 ml/minute (serum creatinine 1.5-5.0 mg/dl) the dosage should be reduced by half (100-200 mg daily). If creatinine clearance is less than 20 ml/minute (serum creatinine greater than 5 mg/dl) 100 mg should be given every 24 hours. In patients undergoing haemodialysis or peritoneal dialysis, 100 mg should be given every 24 hours.
Impaired liver function :
The excretion of ofloxacin may be reduced in patients with severe hepatic dysfunction.
Elderly :
No adjustment of dosage is required in the elderly, other than that imposed by consideration of renal or hepatic function.
Children :
OFLOXACIN TABLETS USP is not indicated for use in children or growing adolescents.
Duration of treatment :
Duration of treatment is dependent on the severity of the infection and the response to treatment. The usual treatment period is 5-10 days except in uncomplicated gonorrhoea, where a single dose is recommended.
CONTRAINDICATIONS :
OFLOXACIN TABLETS USP should not be used in patients with known hypersensitivity to 4-quinolone antibacterials or any of the tablet excipients. OFLOXACIN TABLETS USP should not be used in patients with a past history of tendinitis. Ofloxacin like other 4-quinolones, is contra-indicated in patients with a history of epilepsy or with a lowered seizure threshold. OFLOXACIN TABLETS USP is contra-indicated in children or growing adolescents, and in pregnant or breast-feeding women, since animal experiments do not entirely exclude the risk of damage to the cartilage of joints in the growing subject. Patients with latent or actual defects in glucose-6-phosphate dehydrogenese activity may be prone to haemolytic reactions when treated with quinolone antibacterial agents. OFLOXACIN TABLETS USP contains lactose which is contra-indicated in patients with galactosaemia, the glucose-galactose malabsorption syndrome, or lactase deficiency.
WARNINGS AND PRECAUTIONS :
Ofloxacin is not the drug of first choice for pneumonia caused by Pneumococci or Mycoplama, or angina tonsillaris caused by β-haemolytic Streptococci. Hypersensitivity and allergic reactions have been reported for fluoroquinolones after first administration. Anaphylactic and anaphylactoid reactions can progress to life-threatening shock, even after the first administration. In these cases ofloxacin should be discontinued and suitable treatment (e.g. treatment for shock) should be initiated.
Clostridium difficile-associated disease :
Diarrhoea, particularly if severe, persistent and/or bloody, during or after treatment with ofloxacin, may be symptomatic of pseudo-membranous colitis. If pseudo-membranous colitis is suspected, ofloxacin must be stopped immediately. Appropriate specific antibiotic therapy must be started without delay (e.g. oral vancomycin, oral teicoplanin or metronidazole). Products inhibiting the peristalsis are contraindicated in this clinical situation.
Patients predisposed to seizures :
In case of convulsive seizures, treatment with ofloxacin should be discontinued.
Cardiac Disorders :
Very rare cases of QT interval prolongation have been reported in patients taking fluoroquinolones. Caution should be taken when using fluoroquinolones, including ofloxacin, in patients with known risk factors for prolongation of the QT interval such as, for example :
• congenital long QT syndrome.
• concomitant use of drugs that are known to prolong the QT interval (e.g. Class IA and III anti- arrhythmics, tricyclic antidepressants, macrolides, antipsychotics).
• uncorrected electrolyte imbalance (e.g. hypokalaemia, hypomagnesaemia) cardiac disease (e.g. heart failure, myocardial infarction, bradycardia).
Elderly patients and women may be more sensitive to QTc-prolonging medications. Therefore, caution should be taken when using fluoroquinolones, including Ofloxacin, in these populations. Patients being treated with ofloxacin should not expose themselves
unnecessarily to strong sunlight and should avoid UV rays (sun lamps, solaria).
Patients with history of psychotic disorder :
Psychotic reactions have been reported in patients receiving fluoroquinolones. In some cases these have progressed to suicidal thoughts or self-endangering behaviour including suicide attempt, sometimes after a single dose. In the event that a patient develops these reactions, OFLOXACIN TABLETS USP should be discontinued and appropriate measures instituted. Ofloxacin should be used with caution in patients with a history of psychotic disorder or in patients with psychiatric disease.
Patients with impaired liver function :
OFLOXACIN TABLETS USP should be used with caution in patients with impaired liver function, as liver damage may occur. Cases of fulminant hepatitis potentially leading to liver failure (including fatal cases) have been reported with fluoroquinolones. Patients
should be advised to stop treatment and contact their doctor if signs and symptoms of hepatic disease develop such as anorexia, jaundice, dark urine, pruritis or tender abdomen.
Patients treated with vitamin K antagonists :
Due to possible increase in coagulation tests (PT/INR) and/or bleeding in patients treated with fluoroquinolones, including ofloxacin, in combination with a vitamin K antagonist (e.g.warfarin), coagulation tests should be monitored when these drugs are given concomitantly.
Myasthenia gravis :
OFLOXACIN TABLETS USP should be used with caution in patients with a history of myasthenia gravis. Administration of antibiotics, especially of prolonged, may lead to proliferation of resistant micro-organisms. The patient’s condition must therefore be checked at regular intervals. If a secondary infection occurs, appropriate measures must be taken.
Peripheral neuropathy :
Sensory or sensorimotor peripheral neuropathy has been reported in patients receiving fluoroquinolones, including ofloxacin. OFLOXACIN TABLETS USP should be discontinued if the patient experiences symptoms of neuropathy in order to prevent the development of an irreversible condition.
Hypoglycaemia :
As with all quinolones, hypoglycaemia has been reported, usually in diabetic patients receiving concomitant treatment with an oral hypoglycaemic agent (e.g. glibenclamide) or with insulin. In these diabetic patients, careful monitoring of blood glucose is recommended.
OFLOXACIN TABLETS USP should be used cautiously in diabetic patient.
Patients with glucose-6-phosphate-dehydrogenase deficiency :
Patients with latent or diagnosed glucose-6-phosphate-dehydrogenase deficiency may be predisposed to haemolytic reactions if they are treated with quinolones. OFLOXACIN TABLETS USP should therefore be administered with caution in such patients.
Patients with rare hereditary disorders :
Patients with rare hereditary disorders of galactose intolerance, the Lapp lactase deficiency or glucose-galactose malabsorption should not take this medicine.
Pregnancy : Category C
Based on a limited amount of human data, the use of fluoroquinolones in the first trimester of pregnancy has not been associated with an increased risk of major malformations or other adverse effects on pregnancy outcome. Animal studies have shown damage to the joint cartilage in immature animals but no teratogenic effects. Therefore ofloxacin should not be used during pregnancy.
Nursing Mothers :
In lactating females, a single oral 200-mg dose of ofloxacin resulted in concentrations of ofloxacin in milk that were similar to those found in plasma. Because of the potential for serious adverse reactions from ofloxacin in nursing infants, a decision should be made whether to discontinue nursing or to discontinue the drug, taking into account the importance of the drug to the mother.
Paediatric Use :
Safety and effectiveness in paediatric patients and adolescents below the age of 18 years have not been established. Ofloxacin causes arthropathy (arthrosis) and osteochondrosis in juvenile animals of several species.
EFFECTS ON ABILITY TO DRIVE AND USE MACHINES :
Since there have been occasional reports of somnolence, impairment of skills, dizziness and visual disturbances, patients should know how they react to ofloxacin before they drive or operate machinery. These effects may be enhanced by alcohol.
INTERACTIONS AND INCOMPATIBILITIES :
Antacids, Sucralfate, Metal Cations, Multivitamins :
Quinolones form chelates with alkaline earth and transition metal cations. Administration of quinolones with antacids containing calcium, magnesium, or aluminum, with sucralfate, with divalent or trivalent cations such as iron, or with multivitamins containing zinc or with didanosine, chewable/buffered tablets or the paediatric powder for oral solution may substantially interfere with the absorption of quinolones resulting in systemic levels considerably lower than desired. These agents should not be taken within the two-hour period before or within the two-hour period after ofloxacin administration.
Caffeine :
Interactions between ofloxacin and caffeine have not been detected.
Cimetidine :
Cimetidine has demonstrated interference with the elimination of some quinolones. This interference has resulted in significant increases in half-life and AUC of some quinolones. The potential for interaction between ofloxacin and cimetidine has not been studied.
Cyclosporine :
Elevated serum levels of cyclosporine have been reported with concomitant use of cyclosporine with some other quinolones. The potential for interaction between ofloxacin and cyclosporine has not been studied.
Drugs metabolized by Cytochrome P450 enzymes :
Most quinolone antimicrobial drugs inhibit cytochrome P450 enzyme activity. This may result in a prolonged half-life for some drugs that are also metabolized by this system (e.g., cyclosporine, theophylline/methylxanthines, warfarin) when co-administered with
quinolones. The extent of this inhibition varies among different quinolones.
Non-steroidal anti-inflammatory drugs :
The concomitant administration of a non-steroidal anti-inflammatory drug with a quinolone, including ofloxacin, may increase the risk of CNS stimulation and convulsive seizures.
Probenecid :
The concomitant use of probenecid with certain other quinolones has been reported to affect renal tubular secretion. The effect of probenecid on the elimination of ofloxacin has not been studied.
Theophylline :
Steady-state theophylline levels may increase when ofloxacin and theophylline are administered concurrently. As with other quinolones, concomitant administration of ofloxacin may prolong the half-life of theophylline, elevate serum theophylline levels, and increase the risk of theophylline-related adverse reactions. Theophylline levels should be closely monitored and theophylline dosage adjustments made, if appropriate, when ofloxacin is co-administered. Adverse reactions (including seizures) may occur with or without an elevation in the serum theophylline level.
Warfarin :
Some quinolones have been reported to enhance the effects of the oral anticoagulant warfarin or its derivatives. Therefore, if a quinolone antimicrobial is administered concomitantly with warfarin or its derivatives, the prothrombin time or other suitable coagulation test should be closely monitored.
Antidiabetic agents (e.g., insulin, glyburide/glibenclamide) :
Since disturbances of blood glucose, including hyperglycaemia and hypoglycaemia, have been reported in patients treated concurrently with quinolones and an antidiabetic agent, careful monitoring of blood glucose is recommended when these agents are used concomitantly.
Laboratory tests :
Determination of opiates or porphyrins in urine may give false-positive results during treatment with ofloxacin. It may be necessary to confirm positive opiate or porphyrin screens by more specific methods. Coagulation tests should be monitored in patients treated with vitamin K antagonists because of a possible increase in the effect of coumarin derivatives.
SIDE EFFECTS :
Central Nervous System : Chest pain, headache; dizziness; fatigue; lethargy; drowsiness; insomnia; nervousness.
Dermatological : Rash; pruritus.
Eye, Ear, Nose and Throat : Visual disturbances; transient burning, itching, stinging, inflammation, angioneurotic oedema, urticaria and dermatitis (ophthalmic use).
Gastrointestinal : Diarrhoea; nausea; vomiting; abdominal pain or discomfort; dry or painful mouth; flatulence; dysgeusia.
Genitourinary : Vaginal discharge; genital pruritis.
Hepatic : Increased ALT, AST.
Haematologic : Eosinophilia; lymphocytopenia.
Other : Vaginitis; fever; decreased appetite. Ophthalmic use may possible cause same adverse reactions seen with systemic use because of absorption.
OVERDOSAGE :
The most important signs to be expected following acute overdosage are CNS symptoms such as confusion, dizziness, impairment of consciousness and convulsive seizures as well as gastrointestinal reactions such as nausea and mucosal erosions.
TREATMENT OF OVERDOSAGE :
In the case of overdose steps to remove any unabsorbed ofloxacin e.g. gastric lavage, administration of adsorbants and sodium sulphate, if possible during the first 30 minutes, are recommended; antacids are recommended for protection of the gastric mucosa.
Elimination of ofloxacin may be increased by forced diuresis. In the event of overdose, symptomatic treatment should be implemented. ECG monitoring should be undertaken, because of the possibility of QT interval prolongation.
STORAGE :
Store at controlled room temperature 15°- 30°C (59°- 86°F), protected from moisture and light. Do not refrigerate.
OFLOXACIN TABLETS USP should be stored in a well closed container.
SHELF LIFE :
24 months from the date of manufacture.
PRESENTATION :
OFLOXACIN TABLETS USP contains Ofloxacin USP 200 mg.
1 Blister of 10 Tablets per Box.

 Cardiovascular
Cardiovascular





