
200 mcg/2ml
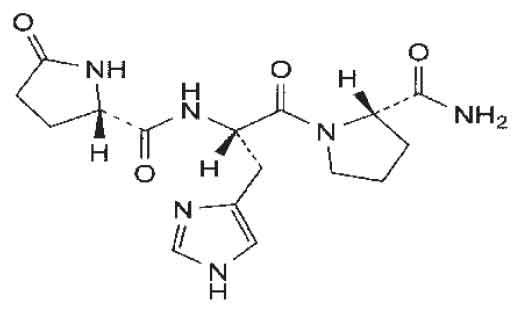
PROTIRELIN Injection (Protirelin) is a synthetic tripeptide that is believed to be structurally identical to the naturally-occurring thyrotropin-releasing hormone produced by the hypothalamus. Chemically, Protirelin is 5-Oxo-L-prolyl-L-histidyl-L-prolinamide. The molecular formula is C16H22N6O4 and molecular weight is 362.4.
STRUCTURAL FORMULA :
Its structural formula is :
PROTIRELIN Injection is a sterile, clear, colourless solution filled in amber ampoule of suitable size.
COMPOSITION :
Each ml contains :
Protirelin B.P. 100 mcg
Methyl Hydroxybenzoate B.P. 1.8 mg
Propyl Hydroxybenzoate B.P. 0.2 mg
(as preservatives)
Sodium Chloride B.P. 9.0 mg
Water for Injections B.P. q.s.
ACTIONS :
Protirelin stimulates the secretion of thyroid stimulating hormone (TSH). Intravenous injection results in a prompt rise in serum TSH levels in normal subjects, peak levels being observed about twenty minutes after administration. There is a concomitant rise in serum levels of prolactin.
PHARMACOKINETICS :
Protirelin intravenous administration, the mean plasma half-life of protirelin in normal subjects is approximately five minutes. TSH levels rise rapidly and reach a peak at 20 to 30 minutes. The decline in TSH levels takes place more slowly, approaching baseline levels after approximately three hours. About 5.5 % of the dose is excreted in the urine, mostly within 30 minutes.
INDICATIONS :
The administration of Protirelin provides a means of assessing thyroid function and the reserve of TSH in the pituitary gland and is recommended as a test procedure where such assessment is indicated.
It is particularly useful as a diagnostic test for :
1. Mild hyperthyroidism
2. Ophthalmic Graves’ disease
3. Mild or preclinical hypothyroidism
4. Hypopituitarism
5. Hypothalamic disease
It may also be used in place of the T3 suppression test.
Administration :
FOR INTRAVENOUS USE.
INSTRUCTIONS FOR USE OF AMPOULE :
The ampoule used in this product is equipped with O.P.C (One Point Cut) opening system. No ampoule file is needed to open the ampoule. The neck of the ampoule is prescored at the point of constriction. A coloured dot on the ampoule head helps to orientate the ampoule. Take the ampoule and face the coloured dot. Let the solution at the head of the ampoule to flow down by shaking or a gentle stroke. The ampoule opens easily by placing the thumb on the coloured dot and gently pressing downwards as shown.

Dosage :
Intravenous injection :
Tests employing intravenous Protirelin are based on the serum TSH response to a standard dose. They provide a means of both quantitative and qualitative assessment of thyroid function. It is essential for each laboratory to establish its own normal range of values for serum TSH before attempting quantitative assessment of Protirelin responses by this means.
Intravenous Protirelin test :
a) Blood sample taken for control TSH assay.
b) Protirelin 200 pg given as a single bolus injection.
c) Blood sample taken 20 minutes after injection for peak TSH assay.
d) If necessary a further blood sample may be taken 60 minutes after injection to detect a delayed TSH response.
The ampoule solution should not be diluted.
The elderly :
The use of Protirelin in the elderly has been well documented. Dosage requirements and the side-effects are similar to those of younger adults. The response may be decreased in elderly subjects, but this does not interfere with the interpretation of the test results.
Children up to the age of 12 :
The procedures for administering Protirelin to children are identical to those outlined above. An intravenous dose of 1 μg/kg bodyweight may be used.
Interpretation of results :
Interpretation of the responses to Protirelin is based on the increase in TSH and/or PBI, T3 or T4 levels from the basal values. In normal subjects, there is a prompt rise in serum levels of TSH. The changes observed in various conditions are briefly outlined below :
1. Hyperthyroidism - no rise in serum TSH or thyroid hormone levels.
2. Ophthalmic Graves’ disease - often no rise in serum TSH or thyroid hormone levels.
3. Primary hypothyroidism - exaggerated and prolonged rise in serum TSH but no change in thyroid hormone levels.
4. Hypopituitarism - absent or impaired TSH or thyroid hormone response implies diminished TSH reserve.
5. Hypothalamic disease - a rise in serum TSH or thyroid hormone levels can occur in the presence of hypothyroidism; delayed responses are common.
The Protirelin test provides, in most instances, information similar to that obtained from a T3 suppression test in that an absent or impaired response usually correlates with an absent or impaired response to T3 suppression.
CONTRAINDICATIONS :
There are no absolute contra-indications to Protirelin.
Transient changes in blood pressure, either increases or decreases, frequently occur immediately following administration of PROTIRELIN Injection. Blood pressure should therefore be measured before PROTIRELIN Injection is administered and at frequent intervals during the first 15 minutes after its administration. Increases in systolic pressure (usually less than 30 mm Hg) and/or increases in diastolic pressure (usually less than 20 mm Hg) have been observed more frequently than decreases in pressure. These changes have not ordinarily persisted for more than 15 minutes nor have they required therapy. More severe degrees of hypertension or hypotension with or without syncope have been reported in a few patients. To minimize the incidence and/or severity of hypotension, the patient should be supine before, during, and after PROTIRELIN Injection administration. If a clinically important change in blood pressure occurs, monitoring of blood pressure should be continued until it returns to base-line levels.
PROTIRELIN Injection should not be administered to patients in whom marked, rapid changes in blood pressure would be dangerous unless the potential benefit clearly outweighs the potential risk. In view of the postulated effect of bolus injections of Protirelin an smooth muscle, patients with bronchial asthma or other types of obstructive airways disease should be closely monitored. Caution should always be observed in patients with myocardial ischaemia and severe hypopituitarism.
PRECAUTIONS :
General :
Thyroid hormones reduce the TSH response to PROTIRELIN Injection. Accordingly, patients in whom PROTIRELIN Injection is to be used diagnostically should be taken off liothyronine (T3) approximately seven days prior to testing and should be taken off thyroid medications containing levothyroxine (T4), e.g., desiccated thyroid, thyroglobulin, or liotrix, at least 14 days before testing. Hormone therapy is NOT to be discontinued when the test is used to evaluate the effectiveness of thyroid suppression with a particular dose of T4 in patients with nodular or diffuse goiter, or for adjustment of thyroid hormone dosage given to patients with primary hypothyroidism. Chronic administration of levodopa has been reported to inhibit the TSH response to PROTIRELIN Injection. It is not advisable to withdraw maintenance doses of adrenocortical drugs used in the therapy of known hypopituitarism. Several published reports have shown that prolonged treatment with glucocorticoids at physiologic doses has no significant effect on the TSH response to thyrotropin releasing hormone, but that the administration of pharmacologic doses of steroids reduces the TSH response. Therapeutic doses of acetylsalicylic acid (2 to 3.6 g/day) have been reported to inhibit the TSH response to protirelin. The ingestion of acetylsalicylic acid caused the peak level of TSH to decrease approximately 30 % as compared to values obtained without acetylsalicylic acid administration. In both cases, the TSH peak occurred 30 minutes post-administration of protirelin.
Pregnancy : Category C
Animal studies have shown an adverse effect and there are no adequate studies in pregnant women OR no animal studies have been conducted and there are no adequate studies in pregnant women.
Nursing mothers :
There are no adequate studies in women for determining infant risk when using this medication during breastfeeding. Weigh the potential benefits against the potential risks before taking this medication while breastfeeding. Breast enlargement and leaking of milk have been reported following the administration of protirelin to lactating women.
Paediatric Use :
Protirelin has been tested in children and, in effective doses, has not been shown to cause different side effects or problems in children than it does in adults.
INTERACTIONS AND INCOMPATIBILITIES :
The secretion of thyrotrophin appears to be modulated by dopaminergic and noradrenergic pathways. The TSH response to Protirelin may be reduced by thyroid hormones, levodopa, phenothiazines, salicylates, bromocriptine, carbamazepine, lithium and by
pharmacological doses of corticosteroids. An increased response may be seen in subjects taking metoclopramide, amiodarone or theophyllines and in men taking oestrogens. Over-treatment with antithyroid drugs may also cause an enhanced response.
SIDE EFFECTS :
Protirelin is well tolerated. Following rapid intravenous injection, side-effects of a mild and transient nature may be experienced. These comprise nausea, a desire to micturate, a feeling of flushing, slight dizziness and a peculiar taste, and have been attributed to a local action of the bolus of Protirelin on the muscle of the gastro-intestinal and genitourinary tracts. A transient increase in pulse rate and blood pressure may also be noted. Pituitary apoplexy requiring acute neurosurgical intervention has been reported infrequently for patients with pituitary macroadenomas following the acute administration of protirelin injection.
OVERDOSAGE :
No symptoms of overdosage have been noted in patients receiving up to 1 mg i.v.
PHARMACEUTICAL PRECAUTIONS :
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit.
STORAGE :
Store below 30°C (86°F), protected from light.
Do not refrigerate.
SHELF LIFE :
24 months from the date of manufacture.
PRESENTATION :
PROTIRELIN Injection is supplied as 200 mcg Protirelin B.P. in 2 ml aqueous solution.
Such 1 Ampoule is packed per Box.
Disclaimer : For the use of a Registered Medical Practitioner or a Hospital or a Institution only. Also it is not intended to be used by healthcare professionals or patients for the purpose of prescribing or administering these products. Questions regarding the complete and current content of product labeling / specification / presentation should be directed to SGPharma.

 Cardiovascular
Cardiovascular





