2.5 mg
For the use of a Registered Medical Practitioner or a Hospital or a Institution only.
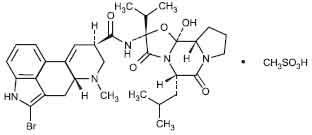
BROMOCRIPTINE MESYLATE TABLETS USP (Bromocriptine Mesylate) is prolactin secretion inhibitor with dopaminergic effect. Chemically, Bromocriptine Mesylate is 2-Bromoergocryptine monomethanesulfonate (salt). The molecular formula is C32H40BrN5O5·CH4SO3 and molecular weight is 750.70.
STRUCTURAL FORMULA :
Its structural formula is :
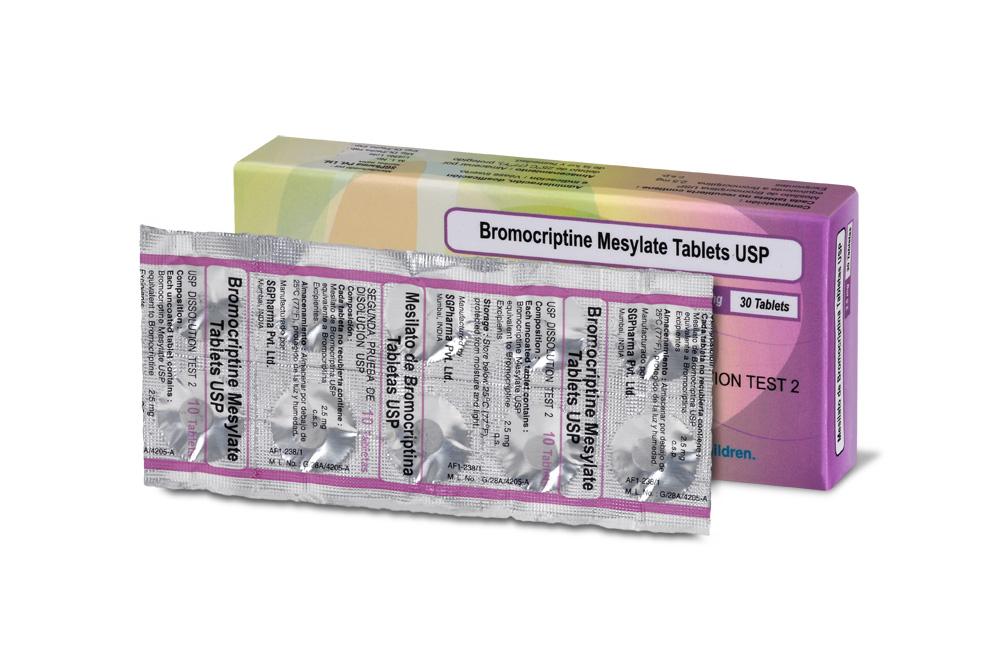
Almost white, circular, flat, uncoated tablets with breakline on one side.
COMPOSITION :
Each uncoated tablet contains :
Bromocriptine Mesylate USP
equivalent to Bromocriptine 2.5 mg
Excipients q.s.
ACTIONS :
Bromocriptine Mesylate is a semi-synthetic ergot alkaloid. This brominated ergot derivative functions as a dopamine D2 receptor agonist and a dopamine D1 receptor antagonist. It imposes a direct dopaminergic effect on cells located within the basal ganglia, mesolimbic
system and hypothalamus. It does not possess the uterotonic and vasoconstrictive properties associated with other ergot preparations. Bromocriptine specifically inhibits the synthesis and secretion of prolactin from the anterior pituitary gland by dopaminergic stimulation
of pituitary prolactin cells. Amenorrhoea, galactorrhoea and other endocrine processes associated with hyperprolactinaemia are consequently returned to physiological levels of activity. Bromocriptine also enhances the release of gonadotrophin and gonadal steroids that are suppressed in hyperprolactinaemia. Preclinical studies have reported that bromocriptine decreases dopamine turnover in the median eminence and dopaminergic tubero-infundibular region of the hypothalamus which may further regulate the synthesis and secretion of prolactin.
Bromocriptine reduces the elevated levels of growth hormone (GH) in acromegaly and may alleviate the clinical symptoms and glucose intolerance presented in this condition. The dopaminemimetic activity of bromocriptine in the striatum may be responsible for the beneficial effects observed in selected cases of Parkinsons Disease.
PHARMACOKINETICS :
Bromocriptine is rapidly absorbed after oral administration, but only 6 % of the dose reaches the systemic circulation due to the high hepatic extraction rate and first pass metabolism. Maximum peak concentrations are obtained within 1 to 1.5 hours; serum prolactin
decreases within 2 hours and is maximally decreased at 8 hours. Bromocriptine is highly distributed in the liver, stomach, and intestine, and plasma protein binding amounts to 96 %. Bromocriptine is extensively metabolised by the liver. The fate of bromocriptine primarily involves biliary excretion with renal excretion of two major metabolites accounting for only 6 % of the total dose. It is not known whether these metabolites (2-bromolysergic acid and 2-bromoisolysergic acid) are pharmacologically active in humans. The elimination of the parent drug from plasma is biphasic, with a terminal half life of about 15 hours (range 8-20 hours). Multiple dosing may result in accumulation of bromocriptine to the extent that plasma levels may be almost double those observed following single doses.
INDICATIONS :
Prolactin dependent Menstrual Cycle disorders (Amenorrhoea, oligomenorrhoea, galactorrhoea), and/or female infertility associated with hyperprolactinaemia or luteal phase deficiency :
Bromocriptine may normalise the menstrual cycle and/or induce ovulation without ovarian overstimulation. Treatment with bromocriptine is not curative and it is not effective in treating ovarian failure.
Pre-menstrual symptoms :
Mood disturbances, bloating, cyclical oedema and breast tenderness.
Hyperprolactinaemia in man :
Prolactin-related hypogonadism (oligospermia, loss of libido, impotence) and galactorrhoea.
Prolactinomas :
Bromocriptine may result in a reduction in size of pituitary prolactinsecreting micro- or macro-adenomas. It can be used alone or prior to radiation or surgery for excision of the tumor. It can also be used post surgery if prolactin levels are still elevated.
Acromegaly :
Used as an adjunct to, or as an alternative to radiation or surgery.
Inhibition of lactation :
Bromocriptine can be used to prevent lactation after an abortion or still-birth, or to suppress puerperal lactation, puerperal breast engorgement & incipient puerperal mastitis. It should not be used to suppress established lactation.
Benign breast disease :
Cyclical mastalgia with pre-menstrual syndrome with or without benign nodular or cystic conditions especially breast disease.
Parkinson’s Disease :
Bromocriptine is used to treat idiopathic or post-encephalitic parkinsonian syndrome either as monotherapy or in combination with other anti-parkinsonion agents.
BROMOCRIPTINE MESYLATE TABLETS USP should always be taken with food.
Prevention or suppression of lactation :
2.5 mg on day 1 (prevention) or daily for 2–3 days (suppression); then 2.5 mg twice daily for 14 days.
Hypogonadism, galactorrhoea, infertility :
Initially 1–1.25 mg at bedtime, increased gradually; usual dose 7.5 mg daily in divided doses, increased if necessary to max. 30 mg daily, usual dose in infertility without hyperprolactinaemia, 2.5 mg twice daily.
Acromegaly : Initially 1-1.25 mg at bedtime, increase gradually to 5 mg every 6 hours.
Prolactinoma : Initially 1-1.25 mg at bedtime; increased gradually to 5 mg every 6 hours (occasional patients may require up to 30 mg daily). Child under 15 years, not recommended.
CONTRAINDICATIONS :
BROMOCRIPTINE MESYLATE TABLETS USP should not be used in post-partum and puerperal women with high blood pressure or coronary artery disease or symptoms and/or a history of serious psychic disorders known malignancy of breast, acute hypersensitivity to bromocriptine or any ergotamine derivative. Very rarely hypertension, myocardial infarction, seizures or stroke (both sometimes preceded by severe headache or visual disturbances), and mental disorders have been reported in postpartum women given bromocriptine for lactation suppression- caution with antihypertensive therapy and avoid other ergot alkaloids. Discontinue immediately if hypertension, unremitting headache, or signs of CNS toxicity develop.
INTERACTIONS AND INCOMPATIBILITIES :
The concomitant administration of erythromycin or other macrolides or octreotide may increase plasma bromocriptine levels. Drugs which can increase prolactin levels, e.g. butyrophenones, phenothiazines, tricyclic antidepressants, reserpine, metoclopramide,
methyldopa, pimozide, oestrogens and TRF, may reduce the efficacy of bromocriptine. Conversely, levodopa, clonidine, pargyline, and iproniazid may synergise the prolactin inhibitory effect of bromocriptine. The hypotensive effects of bromocriptine may be additive with
those of anti-hypertensive agents. Concomitant use of bromocriptine and other ergot alkaloids is not recommended since the combination may cause potentially serious side effects such as myocardial infarction and hypertension. Alcohol may decrease tolerability to bromocriptine.
WARNINGS AND PRECAUTIONS :
Hypotension reactions :
Hypotensive reactions can be disturbing in some patients during the first few days of treatment with bromocriptine. Monitor blood pressure for a few days after starting treatment and following dosage increases; particular care should be exercised when driving or operating machinery; tolerance may be reduced by alcohol.
Fibrotic reactions :
Ergot-derived dopamine-receptor agonists, bromocriptine, cabergoline, and pergolide have been associated with pulmonary, retroperitoneal, and pericardial fibrotic reactions. Exclude cardiac valvulopathy with echocardiography before starting treatment with these ergot derivatives for chronic endocrine disorders (excludes suppression of lactation) or Parkinson’s disease; it may also be appropriate to measure the erythrocyte sedimentation rate and serum creatinine and to obtain a chest X-ray. Patients should be monitored for dyspnoea, persistent cough, chest pain, cardiac failure, and abdominal pain or tenderness. If long term treatment is expected, then lung-function tests may also be helpful.
Psychiatric disorders :
Bromocriptine should be used with caution in patients with a history of dementia or other psychiatric disorders, as high dosages (20 to 40 mg) may be associated with confusion and mental disturbances. May also cause visual or auditory hallucinations alone or combined
with levodopa therapy.
Peptic Ulcer :
Patients with known or suspected peptic ulcers should be treated with caution in respect of several reports of fatal gastric haemorrhage in acromegalic patients who received high doses of bromocriptine. If bromocriptine must be used in acromegalic patients, they should
be instructed to report any gastrointestinal side effects immediately.
Parkinson’s Disease :
Pleural effusions with pleural and pulmonary fibrosis have been observed in long term bromocriptine therapy in patients with Parkinson’s disease. During long term treatment regular monitor with chest x-rays should be considered. The causal relationship between bromocriptine and these signs have not been established, but, patients with unexplained pleuropulmonary signs should be examined thoroughly and discontinuation of treatment considered. Retroperitoneal fibrosis has been reported in a few sufferers of Parkinsons Disease, who received daily doses of more than 30 mg for a number of years. To recognize retoperitoneal fibrosis at an early and reversible stage its manifestations (e.g. Impaired renal function, back pain, and oedema of the lower limbs), be looked for in such patients. Bromocriptine should be withdrawn is fibrotic changes in the peritoneum are diagnosed or suspected.
Diabetic Retinopathy :
Bromocriptine may cause a release of growth hormone in normal and diabetic patients. Growth hormone has been implicated in the acceleration and maintenance of diabetic retinopathy. Bromocriptine should therefore be used with caution in diabetic patients.
Sudden onset of sleep :
Excessive daytime sleepiness and sudden onset of sleep can occur with dopaminergic drugs. Patients starting treatment with these drugs should be warned of the possibility of these effects and ofthe need to exercise caution when driving or operating machinery.
Patients who have suffered excessive sedation or sudden onset of sleep should refrain from driving or operating machines until those effects have stopped recurring. Bromocriptine should be used with caution in patients with impaired liver, renal or severe cardiovascular dysfunction.
Female patients :
Bromocriptine may restore fertility when used in the lowest dose possible to control symptoms, and if pregnancy is not desired during therapy, barrier contraceptive measures should be used. Oral contraceptives are contra-indicated since they may cause amenorrhoea or galactorrhoea. A pregnancy test should be performed every 4 weeks in amenorrhoeic women. If pregnancy occurs, the drug should be discontinued. Although there is no evidence of uterine tumor development in women receiving Bromocriptine, it is recommended that patients on long-term therapy should have regular gynaecological assessments. In patients treated for hyperprolactinaemia, or nodular and/or cystic breast disorders, malignancy of the pituitary or breast, respectively, should be excluded.
Usage in Pregnancy :
Category A
Bromocriptine should be discontinued immediately if pregnancy occurs during therapy, unless there is a definite indication for its continuation. No increased risk of abortion has been observed following the withdrawal of bromocriptine. If patients with prolactinomas show signs of tumor enlargement e.g. headaches and/or visual deterioration following the withdrawal of bromocriptine, they may have therapy re-instituted. In other cases surgery may be appropriate. There is no evidence that the use of bromocriptine is associated with an increased risk of congenital abnormalities.
Use in Lactation :
Bromocriptine inhibits lactation and should not be used by women who elect to breast-feed.
Inhibition of Lactation :
Occasionally serious adverse reactions have been reported. These include seizures, strokes, myocardial infarction, hypertension and psychic disorders. Constant or progressively severe headache, which can be accompanied by visual disturbances, often precede by hours or days the occurrence of a seizure and/or stroke. Periodic monitoring of blood pressure is recommended in postpartum women receiving bromocriptine for the inhibition of lactation. Lactation inhibition therapy should not begin until the vital signs have stabilised and not before 4 hours after delivery, as bromocriptine may cause hypotension or sometimes hypertension in some patients. If hypertension, a severe progressive or unremitting headache (with or without visual disturbance), or the evidence of CNS toxicity develops, the drug therapy should be discontinued and the patient evaluated promptly.
SIDE EFFECTS :
Nausea is the most common side effect at the beginning of therapy with bromocriptine, but vomiting, dizziness, postural hypotension and fainting sometimes occur. Initial doses must be low, taken preferably at bedtime, and blood pressure should be monitored initially.
Gastrointestinal effects include abdominal cramps, epigastric pain, indigestion, constipation or diarrhoea. Occasionally, acromegalic patients on doses 10 to 60 mg bromocriptine have developed a peptic ulcer or gastrointestinal haemorrhage. Temporary reduction of dose or administration with food may relieve these effects. Additional side-effects include headache, nasal congestion or watery rhinorrhoea, dryness of mouth, and drowsiness, Dyskinesias have occurred in patients with Parkinsonism as well as psychosis, nightmares,
anxiety, mania, hallucinations, confusion and erythromelalgia. These are more likely at higher doses, but may also occur at low doses. Mania has also been reported when bromocriptine has been used post-partum. Episodes of reversible vasoconstriction in the
extremities induced by cold, and leg cramps have been reported during prolonged therapy or with high doses.
Other adverse effects reported include depression, anxiety and extreme agitation, paraesthesias, fatigue, arrhythmias or exacerbation of angina. Pleuropulmonary changes and retroperitoneal fibrosis have occurred in patients on long-term therapy. (see Warnings and Precautions). In several acromegalic patients treated with high doses, fatal gastric haemorrhage has been reported. (See Warnings and Precautions). The use of bromocriptine for the inhibition of physiological lactation postpartum has been associated with the rare occurrence of hypertension, myocardial infarction, seizures, strokes and psychiatric disorders. (See Contraindications and Warnings and Precautions).
OVERDOSAGE AND TREATMENT OF OVERDOSAGE :
Overdosage of BROMOCRIPTINE MESYLATE TABLETS USP may cause nausea, vomitting, dizziness, postural hypotension, sweating, drowsiness and hallucinations. The management of acute intoxication is symptomatic. Metoclopramide may be indicated for the treatment of emesis or hallucinations.
STORAGE :
Store below 30°C (86°F), protected from moisture and light.
Do not refrigerate.
PRESENTATION :
3 Strips of 10 Tablets per Box.
Disclaimer : For the use of a Registered Medical Practitioner or a Hospital or a Institution only. Also it is not intended to be used by healthcare professionals or patients for the purpose of prescribing or administering these products. Questions regarding the complete and current content of product labeling / specification / presentation should be directed to SGPharma.

 Cardiovascular
Cardiovascular