125 mg
For the use of a Registered Medical Practitioner or a Hospital or a Institution only.
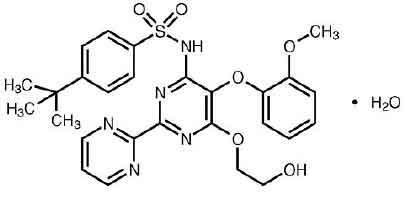
BOSENTAN TABLETS (Bosentan Monohydrate) is an endothelin receptor antagonist. Chemically, Bosentan Monohydrate is designated as 4-tertbutyl-N-[6-(2-hydroxy-ethoxy)-5-(2-methoxy-phenoxy)-[2,2’]-bipyrimidin-4-yl]-benzenesulphonamide monohydrate. The molecular formula is C27H29N5O6S.H2O and molecular weight is 569.64.
STRUCTURAL FORMULA :
Its structural formula is :
BOSENTAN TABLETS are orange coloured, circular, biconvex film coated tablets.
COMPOSITION :
Each film coated tablet contains :
Bosentan Monohydrate
equivalent to Bosentan 125 mg
Excipients q.s.
Colours : Yellow Oxide of Iron, Red Oxide of Iron & Titanium Dioxide B.P.
ACTIONS :
Bosentan is a specific and competitive antagonist at endothelin receptor types ETA and ETB. Bosentan has a slightly higher affinity for ETA receptors than for ETB receptors. The clinical impact of dual endothelin blockage is unknown. Endothelin-1 (ET-1) is a neurohormone, the effects of which are mediated by binding to ETA and ETA receptors in the endothelium and vascular smooth muscle. ET-1 concentrations are elevated in plasma and lung tissue of patients with pulmonary arterial hypertension, suggesting a pathogenic role for ET-1 in this disease.
PHARMACOKINETICS :
Bosentan is absorbed from the gastrointestinal tract with an absolute bioavailability of about 50 %. Peak plasma concentrations occur about 3 to 5 hours after an oral dose. It is more than 98 % bound to plasma proteins, mainly to albumin. Bosentan is metabolised in the liver by the cytochrome P450 isoenzymes CYP2C9 and CYP3A4 and is an inducer of these enzymes and possibly also of CYP2C19; after multiple dosing, plasma concentrations of bosentan decrease gradually to 50 to 65 % of those seen after a single dose.
Bosentan has three metabolites, one of which is active. Bosentan is excreted almost entirely as metabolites in the bile; less than 3 % of an oral dose is excreted in the urine. The terminal elimination half-life is about 5 hours.
INDICATIONS :
Treatment of pulmonary arterial hypertension (PAH) to improve exercise capacity and symptoms in patients with WHO functional class III. Efficacy has been shown in :
- Primary (idiopathic and heritable) pulmonary arterial hypertension.
- Pulmonary arterial hypertension secondary to scleroderma without significant interstitial pulmonary disease.
- Pulmonary arterial hypertension associated with congenital systemic-to-pulmonary shunts and Eisenmenger’s physiology.
- Some improvements have also been shown in patients with pulmonary arterial hypertension WHO functional class II.
BOSENTAN TABLETS is also indicated to reduce the number of new digital ulcers in patients with systemic sclerosis and ongoing digital ulcer disease.
Administration :
BOSENTAN TABLETS are for oral administration. BOSENTAN TABLETS are to be taken orally morning and evening, with or without food. The film-coated tablets are to be swallowed with water.
Dosage :
Pulmonary arterial hypertension :
Treatment should only be initiated and monitored by a physician experienced in the treatment of pulmonary arterial hypertension.
Adults :
In adult patients, BOSENTAN TABLETS treatment should be initiated at a dose of 62.5 mg twice daily for 4 weeks and then increased to the maintenance dose of 125 mg twice daily. The same recommendations apply to re-introduction of BOSENTAN TABLETS after treatment interruption.
Paediatric population :
In children, the optimal maintenance dose has not been defined in well-controlled studies. However, paediatric pharmacokinetic data have shown that bosentan plasma concentrations in children were on average lower than in adult patients and were not increased by increasing the dose of BOSENTAN TABLETS above 2 mg/kg body weight twice daily. Based on these pharmacokinetic results, higher doses are unlikely to be more effective, and greater adverse reaction rates cannot formally be excluded in young children if the dose is increased. When used in children 2 years and older, the recommended starting and maintenance dose is therefore 2 mg/kg morning and evening. No clinical study has been conducted to compare the efficacy/safety ratio of 2 mg/kg to 4 mg/kg body weight twice daily in children. There is only limited clinical experience in paediatric patients under 2 years of age.
Management in case of clinical deterioration of PAH :
In the case of clinical deterioration (e.g., decrease in 6-minute walk test distance by at least 10 % compared with pre-treatment measurement) despite BOSENTAN TABLETS treatment for at least 8 weeks (target dose for at least 4 weeks), alternative therapies should be considered. However, some patients who show no response after 8 weeks of treatment with BOSENTAN TABLETS may respond favourably after an additional 4 to 8 weeks of treatment. In the case of late clinical deterioration despite treatment with BOSENTAN TABLETS (i.e., after several months of treatment), the treatment should be re-assessed. Some patients not responding well to 125 mg twice daily of BOSENTAN TABLETS may slightly improve their exercise capacity when the dose is increased to 250 mg twice daily. A careful benefit/risk assessment should be made, taking into consideration that the liver toxicity is dose dependent.
Discontinuation of treatment :
There is limited experience with abrupt discontinuation of BOSENTAN TABLETS in patients with pulmonary arterial hypertension. No evidence for acute rebound has been observed. However, to avoid the possible occurrence of harmful clinical deterioration due to potential rebound effect, gradual dose reduction (halving the dose for 3 to 7 days) should be considered. Intensified monitoring is recommended during the discontinuation period. If the decision to withdraw BOSENTAN TABLETS is taken, it should be done gradually while an alternative therapy is introduced.
Systemic sclerosis with ongoing digital ulcer disease :
Treatment should only be initiated and monitored by a physician experienced in the treatment of systemic sclerosis.
Adults :
BOSENTAN TABLETS treatment should be initiated at a dose of 62.5 mg twice daily for 4 weeks and then increased to the maintenance dose of 125 mg twice daily. The same recommendations apply to re-introduction of BOSENTAN TABLETS after treatment interruption. Controlled clinical study experience in this indication is limited to 6 months. The patient’s response to treatment and need for continued therapy should be re-evaluated on a regular basis. A careful benefit/risk assessment should be made, taking into consideration the liver toxicity of bosentan.
Paediatric population :
There are no data on the safety and efficacy in patients under the age of 18 years. Pharmacokinetic data are not available for BOSENTAN TABLETS in young children with this disease.
Special populations :
Patients with hepatic impairment :
BOSENTAN TABLETS is contraindicated in patients with moderate to severe liver dysfunction. No dose adjustment is needed in patients with mild hepatic impairment (i.e., Child-Pugh class A).
Patients with renal impairment :
No dose adjustment is required in patients with renal impairment. No dose adjustment is required in patients undergoing dialysis.
Elderly population :
No dose adjustment is required in patients over the age of 65 years.
CONTRAINDICATIONS :
- Hypersensitivity to the active substance or to any of the excipients.
- Moderate to severe hepatic impairment, i.e., Child-Pugh class B or C.
- Baseline values of liver aminotransferases, i.e., aspartate aminotransferases (AST) and/or alanine aminotransferases (ALT), greater than 3 times the upper limit of normal.
- Concomitant use of cyclosporine A.
- Pregnancy.
- Women of child-bearing potential who are not using reliable methods of contraception.
Potential Liver Injury :
Elevations in ALT or AST by more than 3 × ULN were observed in 11 % of bosentan-treated patients (N = 658) compared to 2 % of placebo-treated patients (N = 280). Three-fold increases were seen in 12 % of 95 PAH patients on 125 mg b.i.d. and 14 % of 70 PAH patients on 250 mg b.i.d. Eightfold increases were seen in 2 % of PAH patients on 125 mg b.i.d. and 7 % of PAH patients on 250 mg b.i.d. Bilirubin increases to >/= 3 × ULN were associated with aminotransferase increases in 2 of 658 (0.3 %) of patients treated with BOSENTAN TABLETS. The combination of hepatocellular injury (increases in aminotransferases of > 3 × ULN) and increases in total bilirubin (>/= 3 × ULN) is a marker for potential serious liver injury.
Elevations of AST and/or ALT associated with bosentan are dose-dependent, occur both early and late in treatment, usually progress slowly, are typically asymptomatic, and to date have been reversible after treatment interruption or cessation. These aminotransferase elevations may reverse spontaneously while continuing treatment with BOSENTAN TABLETS. Liver aminotransferase levels must be measured prior to initiation of treatment and then monthly. If elevated aminotransferase levels are seen, changes in monitoring and treatment must be initiated. If liver aminotransferase elevations are accompanied by clinical symptoms of liver injury (such as nausea, vomiting, fever, abdominal pain, jaundice, or unusual lethargy or fatigue) or increases in bilirubin >/= 2 × ULN, treatment should be
stopped. There is no experience with the re-introduction of BOSENTAN TABLETS in these circumstances.
Pre-existing Liver Impairment :
Liver aminotransferase levels must be measured prior to initiation of treatment and then monthly. BOSENTAN TABLETS should generally be avoided in patients with moderate or severe liver impairment. In addition, BOSENTAN TABLETS should generally be avoided in patients with elevated aminotransferases (> 3 × ULN) because monitoring liver injury in these patients may be more difficult.
PRECAUTIONS :
Haematologic Changes :
Treatment with BOSENTAN TABLETS caused a dose-related decrease in haemoglobin and haematocrit. Haemoglobin levels should be monitored after 1 and 3 months of treatment and then every 3 months. The overall mean decrease in haemoglobin concentration for
bosentan-treated patients was 0.9 gm/dl (change to end of treatment). Most of this decrease of haemoglobin concentration was detected during the first few weeks of bosentan treatment and haemoglobin levels stabilized by 4 - 12 weeks of bosentan treatment. In placebo-controlled studies of all uses of bosentan, marked decreases in haemoglobin (> 15 % decrease from baseline resulting in values < 11 gm/dl) were observed in 6 % of bosentan treated patients and 3 % of placebo-treated patients. In patients with pulmonary arterial hypertension treated with doses of 125 and 250 mg b.i.d., marked decreases in haemoglobin occurred in 3 % compared to 1 % in placebo-treated patients. A decrease in haemoglobin concentration by at least 1 gm/dl was observed in 57 % of bosentan-treated patients as compared to 29 % of placebo-treated patients. In 80 % of those patients whose haemoglobin decreased by at least 1 gm/dl, the decrease occurred during the first 6 weeks of bosentan treatment.
During the course of treatment the haemoglobin concentration remained within normal limits in 68 % of bosentan - treated patients compared to 76 % of placebo patients. The explanation for the change in haemoglobin is not known, but it does not appear to be haemorrhage or haemolysis. It is recommended that haemoglobin concentrations be checked after 1 and 3 months, and every 3 months thereafter. If a marked decrease in haemoglobin concentration occurs, further evaluation should be undertaken to determine the cause and need for specific treatment.
Fluid retention :
In a placebo-controlled trial of patients with severe chronic heart failure, there was an increased incidence of hospitalization for CHF associated with weight gain and increased leg oedema during the first 4-8 weeks of treatment with BOSENTAN TABLETS. In addition, there have been numerous postmarketing reports of fluid retention in patients with pulmonary hypertension, occurring within weeks after starting BOSENTAN TABLETS. Patients required intervention with a diuretic, fluid management, or hospitalization for
decompensating heart failure.
Pregnancy : Category X
Studies in animals have shown reproductive toxicity (teratogenicity, embryotoxicity). There are no reliable data on the use of BOSENTAN TABLETS in pregnant women. The potential risk for humans is still unknown. BOSENTAN TABLETS is contraindicated in pregnancy.
Use in women of child-bearing potential :
Before the initiation of BOSENTAN TABLETS treatment in women of child-bearing potential, the absence of pregnancy should be checked, appropriate advice on reliable methods of contraception provided, and reliable contraception initiated. Patients and prescribers must be aware that due to potential pharmacokinetic interactions, BOSENTAN TABLETS may render hormonal contraceptives ineffective. Therefore, women of child-bearing potential must not use hormonal contraceptives (including oral, injectable, transdermal or
implantable forms) as the sole method of contraception but must use an additional or an alternative reliable method of contraception. If there is any doubt about what contraceptive advice should be given to the individual patient, consultation with a gynaecologist is recommended. Because of possible hormonal contraception failure during BOSENTAN TABLETS treatment, and also bearing in mind the risk that pulmonary hypertension severely deteriorates with pregnancy, monthly pregnancy tests during treatment with BOSENTAN TABLETS are recommendedto allow early detection of pregnancy.
Nursing Mothers :
It is not known whether bosentan is excreted into human breast milk. Breast-feeding is not recommended during treatment with BOSENTAN TABLETS.
Paediatric Use :
Safety and efficacy in paediatric patients have not been established.
EFFECTS ON ABILITY TO DRIVE AND USE MACHINES :
No specific studies have been conducted to assess the direct effect of BOSENTAN TABLETS on the ability to drive and use machines. However, BOSENTAN TABLETS may induce hypotension, with symptoms of dizziness or syncope that could affect the ability to drive or use machines.
INTERACTIONS :
Cytochrome P450 Summary :
Bosentan is metabolized by CYP2C9 and CYP3A. Inhibition of these enzymes may increase the plasma concentration of bosentan. Concomitant administration of both a CYP2C9 inhibitor (such as fluconazole or amiodarone) and a strong CYP3A inhibitor (e.g., ketoconazole, itraconazole) or a moderate CYP3A inhibitor (e.g., amprenavir, erythromycin, fluconazole, diltiazem) with bosentan will likely lead to large increases in plasma concentrations of bosentan. Coadministration of such combinations of a CYP2C9 inhibitor plus a strong or moderate CYP3A inhibitor with BOSENTAN TABLETS is not recommended. Bosentan is an inducer of CYP3A and CYP2C9. Consequently plasma concentrations of drugs metabolized by these two isozymes will be decreased when BOSENTAN TABLETS is coadministered. Bosentan had no relevant inhibitory effect on any CYP isozyme in vitro (CYP1A2, CYP2C9, CYP2C19, CYP2D6, CYP3A). Consequently, BOSENTAN TABLETS is not expected to increase the plasma concentrations of drugs metabolized by these enzymes.
Hormonal Contraceptives :
Hormonal contraceptives, including oral, injectable, transdermal, and implantable forms, may not be reliable when BOSENTAN TABLETS is coadministered. Females should practice additional methods of contraception and not rely on hormonal contraception alone when taking BOSENTAN TABLETS. An interaction study demonstrated that coadministration of bosentan and a combination oral hormonal contraceptive produced average decreases of norethindrone and ethinyl estradiol levels of 14 % and 31 %, respectively. However, decreases in exposure were as much as 56 % and 66 %, respectively, in individual subjects.
SIDE EFFECTS :
Gastro-intestinal disturbances, dry mouth, rectal haemorrhage, hepatic impairment, flushing, hypotension, palpitation, oedema, chest pain; dyspnoea; headache, dizziness, fatigue; back pain and pain in extremities; anaemia; hypersensitivity reactions (including rash, pruritus, and anaphylaxis).
INFORMATION FOR PATIENTS :
Patients are advised to consult the BOSENTAN TABLETS Medication Guide on the safe use of BOSENTAN TABLETS. The physician should discuss with the patient the importance of monthly monitoring of serum aminotransferases and urine or serum pregnancy testing and of avoidance of pregnancy. The physician should discuss options for effective contraception and measures to prevent pregnancy with their female patients. Input from a gynaecologist or similar expert on adequate contraception should be sought as needed.
OVERDOSAGE :
Bosentan has been given as a single dose of up to 2400 mg in normal volunteers, or up to 2000 mg/day for 2 months in patients, without any major clinical consequences. The most common side effect was headache of mild to moderate intensity. In the cyclosporine A interaction study, in which doses of 500 and 1000 mg b.i.d. of bosentan were given concomitantly with cyclosporine A, trough plasma concentrations of bosentan increased 30-fold, resulting in severe headache, nausea, and vomiting, but no serious adverse events. Mild decreases in blood pressure and increases in heart rate were observed.
TREATMENT OF OVERDOSAGE :
There is no specific experience of overdosage with bosentan beyond the doses described above. Massive overdosage may result in pronounced hypotension requiring active cardiovascular support.
STORAGE :
Store below 30°C (86°F), protected from moisture and light.
Do not refrigerate.
SHELF LIFE :
24 months from the date of manufacture.
PRESENTATION :
BOSENTAN TABLETS contains Bosentan Monohydrate equivalent to Bosentan 125 mg.
3 Strips of 10 Tablets per Box.
Disclaimer : For the use of a Registered Medical Practitioner or a Hospital or a Institution only. Also it is not intended to be used by healthcare professionals or patients for the purpose of prescribing or administering these products. Questions regarding the complete and current content of product labeling / specification / presentation should be directed to SGPharma.

 Cardiovascular
Cardiovascular