250 mg/5 ml, 500 mg/10 ml
For the use of a Registered Medical Practitioner or a Hospital or a Institution only.
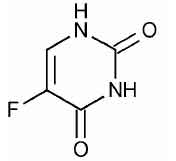
Fluorouracil is an analogue of uracil, a component of ribonucleic acid and is an antimetabolite. Chemically, Fluorouracil is 2,4(1H,3H)–Pyrimidinedione, 5-fluoro-It’s molecular formula is C4H3FN2O2 and molecular weight is 130.08.
STRUCTURAL FORMULA :
Its structural formula is :
Fluorouracil Injection is a sterile, clear, colourless or slightly yellow solution filled in vials of suitable size.
COMPOSITION :
Each ml contains :
Fluorouracil USP 50 mg
Water for Injection USP q.s.
Contains no preservatives.
ACTIONS :
Fluorouracil is an analogue of uracil, a component of ribonucleic acid. The drug is believed to function as an antimetabolite. After intracellular conversion to the active deoxynucleotide, it interferes with the synthesis of DNA by blocking the conversion of deoxyuridylic acid to thymidylic acid by the cellular enzyme thymidylate synthetase. Fluorouracil may also interfere with RNA synthesis.
PHARMACOKINETICS :
After intravenous administration, Fluorouracil is distributed through the body water and disappears from the blood within 3 hours. It is preferentially taken up by actively dividing tissues and tumours after conversion to its nucleotide. Fluorouracil readily enters the C.S.F and brain tissue. Following I.V. administration, the plasma elimination half-life averages about 16 minutes and is dose dependant. Following a single intravenous dose of Fluorouracil approximately 15 % of the dose is excreted unchanged in the urine within 6 hours; over 90 % of this is excreted in the first hour. The remainder is mostly metabolised in the liver by the usual body mechanisms for uracil.
INDICATIONS :
For the palliative treatment, either alone or in combination of malignant tumours particularly of the breast, colon and rectum. It is also used for the treatment of gastric, primary hepatic, pancreatic, uterine, ovarian and bladder carcinomas.
Breast cancer :
Fluorouracil has been used as part of combination therapy as an adjunct to surgery in the treatment of early breast cancer in women with negative auxiliary lymph nodes and oestrogen receptor negative tumours. Fluorouracil has also been used to treat more advanced forms of breast cancer including inoperable cancer.
Gastrointestinal cancer :
Fluorouracil, when used as an adjunct to surgery, has produced temporary improvement in a substantial number of patients with advanced carcinoma of the gastrointestinal tract.
Administration :
Fluorouracil Injection can be given by Intravenous Injection or Intravenous or Intra-arterial Infusion.
Adults :
Selection of an appropriate dose and treatment regime depends upon the condition of the patient, the type of carcinoma being treated and whether Fluorouracil is to be administered alone or in combination with other therapy. Initial treatment should be given in hospital and
the total daily dose should not exceed 1g. It is customary to calculate the dose in accordance with the patient’s actual bodyweight unless there is obesity, oedema or some other form of abnormal fluid retention such as ascites. In this case, ideal weight is used as the basis for
calculation.
Reduction of the dose is advisable in patients with any of the following :
1. Cachexia.
2. Major surgery within preceding 30 days.
3. Reduced bone marrow function.
4. Impaired hepatic or renal function.
Dosage :
Adult Dose :
The following regimen have been recommended for use as a single agent.
Initial Treatment :
This may be in the form of an infusion or an injection, the former usually being preferred because of lesser toxicity.
Intravenous Infusion :
15 mg/kg bodyweight but not more than 1 g per infusion, diluted in 500 ml of 5 % Glucose or 0.9 % Sodium Chloride Injection and given by intravenous infusion at a rate of 40 drops per minute over 4 hours. Alternatively the daily dose may be infused over 30 - 60 minutes or may be given as a continuous infusion over 24 hours. The infusion may be repeated daily until there is evidence of toxicity or a total dose of 12 - 15 g has been reached.
Intravenous Injection :
12 mg/kg bodyweight may be given daily for 3 days and then, if there is no evidence of toxicity, 6 mg/kg on alternate days for 3 further doses. An alternative regime is 15 mg/kg as a single intravenous injection once a week throughout the course.
Intra-arterial Infusion :
5 - 7.5 mg/kg bodyweight daily may be given by 24 hour continuous intra-arterial infusion.
Maintenance Therapy :
An initial intensive course may be followed by maintenance therapy providing there are no significant toxic effects. In all instances, toxic side effects must disappear before maintenance therapy is started.
The initial course of Fluorouracil can be repeated after an interval of 4 to 6 weeks from the last dose or alternatively, treatment can be continued with intravenous injections of 5 - 15 mg/kg bodyweight at weekly intervals. This sequence constitutes a course of therapy. Some patients have received up to 30 g at a maximum rate of 1g daily. A more recent alternative method is to give 15 mg/kg I.V. once a week throughout the course of treatment. This obviates the need for an initial period of daily administration.
In combination with Irradiation :
Irradiation combined with Fluorouracil has been found to be useful in the treatment of certain types of metastatic lesions in the lungs and for the relief of pain caused by recurrent, inoperable growth. The standard dose of Fluorouracil should be used.
CONTRAINDICATIONS :
Fluorouracil is contraindicated in seriously debilitated patients or those with bone marrow depression after radiotherapy or treatment with other antineoplastic agents. Fluorouracil is strictly contraindicated in pregnant or breast feeding women. Fluorouracil should not be used in the management of non- malignant disease.
WARNINGS :
It is recommended that Fluorouracil be given only by or under strict supervision of a qualified physician who is well acquainted with the use of potent antimetabolites. Because of the possibility of severe toxic reactions, all patients should be hospitalised, at least during the initial course of therapy. Fluorouracil should not be readministered after a documented cardiovascular reaction (arrhythmia, angina, ST segment changes) as there is a risk of sudden death. Any form of therapy which adds to the stress of the patient, interferes with nutritional uptake or depresses bone marrow function, will increase the toxicity of Fluorouracil.
PRECAUTIONS :
Adequate treatment with Fluorouracil is usually followed by leucopenia, the lowest white blood cell (W.B.C.) count commonly being observed between the 9th and 14th day of the first course, but occasionally being delayed for as long as 20 days. The count usually returns to normal by the 30th day. Daily monitoring of platelets and W.B.C. counts are recommended and treatment should be stopped if platelets fall below 100,000 per mm³ or the W.B.C. count falls below 3,500 per mm³. If the W.B.C. count falls below 2,000 per mm³, it is recommended that the patient be placed in protective isolation in the hospital and given the appropriate preventative treatment for systemic infection. Treatment should also be discontinued at the first sign of oral ulceration or if there is evidence of gastrointestinal side effects such as stomatitis, diarrhoea or bleeding from the G.I. tract or haemorrhage at any site. The ratio between effective and toxic dose is small and therapeutic response is unlikely without some degree of toxicity. Care must be taken, therefore, in the selection of
patients and adjustment of dosage. Fluorouracil should be used with caution in patients with reduced renal or liver function or jaundice. Fluorouracil should be used with caution in elderly patients. Age 70 years or older and the female gender are reported independent risk factors for severe toxicity from Fluorouracil based chemotherapy. Close monitoring for multiple organ toxicities and vigorous supportive care of those with toxicity are necessary. There have been reports of increased toxicity in patients who have reduced activity/deficiency of the enzyme dihydropyrimidine dehydrogenase (DPD).
Pregnancy Category D
Fluorouracil is strictly contraindicated in pregnant and breast feeding women.
INTERACTIONS :
Leucovorin (folinic acid) enhances the DNA-directed toxicity of Fluorouracil. This combination should be used with caution as it is reported to increase the gastrointestinal toxicity of Fluorouracil. Allopurinol may decrease the degree of bone marrow depression produced by Fluorouracil. Various agents have been reported to biochemically modulate the antitumour efficacy or toxicity of Fluorouracil. Common drugs include methotrexate, metronidazole and leucovorin. Pre-treatment with cimetidine prior to intravenous Fluorouracil increased the area under the curve by 27 %. The total body clearance was reduced by 28 %.
COMPATIBILITIES :
1. Fluorouracil Injection when admixed with 0.9 % Sodium Chloride Injection has been shown to retain at least 95 % of initial potency over a 7 days period stored at room temperature and in the presence of fluorescent light.
2. Fluorouracil Injection when admixed with 5 % Dextrose Injection has been shown to retain at least 95 % of initial potency over a 5 days period stored at room temperature and in the presence of fluorescent light.
INCOMPATIBILITIES :
Admixture with acidic drugs or drugs that decompose in an alkaline environment should be avoided. Fluorouracil is incompatible with Carboplatin, Cisplatin, Cytarabine, Diazepam, Doxorubicin, other Anthracyclines and possibly Methotrexate.
SIDE EFFECTS :
Diarrhoea, nausea and vomiting are seen commonly during therapy and may be treated symptomatically. Nausea and vomiting may be controlled with an appropriate antiemetic. Alopecia may be seen in a substantial number of cases, but is reversible. Dermatitis,
pigmentation, changes in the nails, ataxia and fever have also been reported. Leucopenia and thrombocytopenia are often encountered during the initial treatment, but may be less frequent during maintenance therapy. There have been reports of chest pain, tachycardia, breathlessness, arrhythmia and E.C.G. changes (ST segment changes) after administration of Fluorouracil. There have been reports of sudden death in patients readministered Fluorouracil after a documented cardiovascular reaction. Systemic Fluorouracil treatment has been associated with various types of ocular toxicity. Additionally several other reports have been noted including excessive lacrimation, dacryostenosis, visual changes and photophobia. Palmar-Plantar Erythrodysesthesia Syndrome, thrombophlebitis and asymptomatic
hyperpigmentation over vascular channels have also been reported. Disorientation, confusion, euphoria, ataxia, nystagmus, headache, slurred speech, dizziness, unsteadiness and acute cerebellar syndrome have occurred in patients receiving fluorouracil. These symptoms may persist after therapy is discontinued.
OVERDOSAGES AND TREATMENT OF OVERDOSAGES :
The symptoms and signs of overdosage are qualitatively similar to the adverse reactions and should be managed as indicated under “Side effects”, “Warnings” and “Precautions”.
PHARMACEUTICAL PRECAUTIONS :
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit. If a precipitate is formed as a result of a exposure to low temperature, redisslove it by heating to 60° with vigorous shaking and allow to cool to body temperature prior to use. The product should be discarded if it appears brown or dark yellow in solution.
SPECIAL PRECAUTIONS FOR DISPOSAL AND OTHER HANDLING :
Fluorouracil Injection should be administered only by or under the direct supervision of a qualified physician who is experienced in the use of cancer chemotherapeutic agents. Fluorouracil Injection should only be prepared for administration by professionals who have been trained in the safe use of the preparation. Preparation should only be carried out in an aseptic cabinet or suite dedicated for the assembly of cytotoxics. In the event of spillage, operators should put on gloves, face mask, eye protection and disposable apron and mop up the spilled material with a absorbent material kept in the area for that purpose. The area should then be cleaned and all contaminated material transferred to a cytotoxic spillage bag or bin and sealed for incineration.
Contamination :
Fluorouracil is an irritant, contact with skin and mucous membranes should be avoided. In the event of contact with the skin or eyes, the affected area should be washed with copious amounts of water or normal saline. A bland cream may be used to treat the transient stinging of the skin. Medical advice should be sought if the eyes are affected or if the preparation is inhaled or ingested.
Preparation Guidelines :
a) Chemotherapeutic agents should be prepared for administration only by professionals who have been trained in the safe use of the preparation.
b) Operations such as reconstitution of powder and transfer to syringes should be carried out only under aseptic conditions in a suite or cabinet dedicated for the assembly of cytotoxics.
c) The personnel carrying out these procedures should be adequately protected with clothing, gloves and eye shield.
d) Pregnant personnel are advised not to handle chemotherapeutic agents.
Disposal:
All materials that have been utilised for dilution and administration should be disposed off according to standard procedures (incineration).
Diluents:
Fluorouracil may be diluted with 5 % glucose or 0.9 % sodium chloride intravenous infusions immediately before parenteral use. The remainder of solutions should be discarded after use; do not make up into multi-dose preparations.
STORAGE :
Store at controlled room temperature 15°-30°C (59°- 86°F), protected from light.
Do not refrigerate.
SHELF LIFE :
24 months from the date of manufacture.
PRESENTATION :
Fluorouracil Injection is supplied as below table :
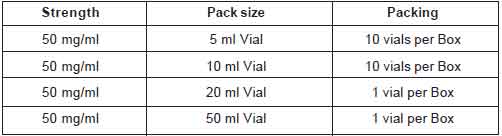
Disclaimer : For the use of a Registered Medical Practitioner or a Hospital or a Institution only. Also it is not intended to be used by healthcare professionals or patients for the purpose of prescribing or administering these products. Questions regarding the complete and current content of product labeling / specification / presentation should be directed to SGPharma.

 Cardiovascular
Cardiovascular