50 mg
For the use of a Registered Medical Practitioner or a Hospital or a Institution only.
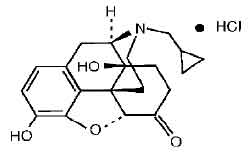
Naltrexone Hydrochloride, an opioid antagonist, is a synthetic congener of oxymorphone with no opioid agonist properties. Naltrexone differs in structure from oxymorphone in that the methyl group on the nitrogen atom is replaced by a cyclopropylmethyl group. Naltrexone is also related to the potent opioid antagonist, naloxone, or n-allylnoroxymorphone. Chemically Naltrexone Hydrochloride is 17-(Cyclopropylmethyl)-4,5α-epoxy-3,14 dihydroxymorphinan-6-one hydrochloride.The molecular formula is C20H23NO4·HCl and molecular weight is 377.86
STRUCTURAL FORMULA :
Its structural formula is :
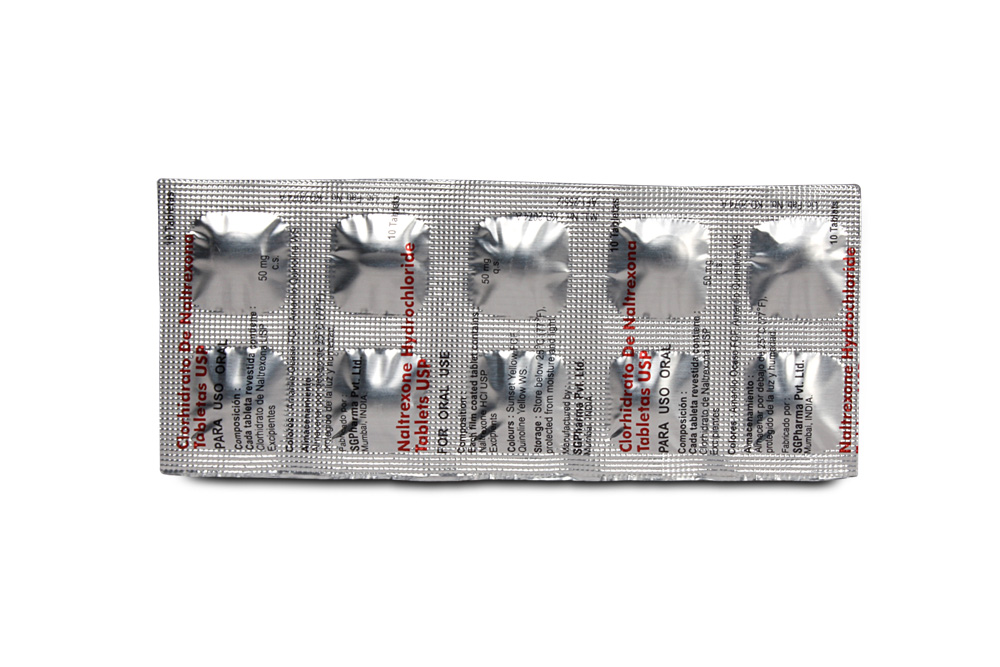
Naltrexone Hydrochloride Tablets are orange coloured, circular, biconvex film coated tablets.
COMPOSITION :
Each film coated tablet contains :
Naltrexone HCl USP 50 mg
Excipients q.s.
Colours : Sunset Yellow FCF, Quinoline Yellow WS
ACTIONS :
Naltrexone is a pure opioid antagonist. It markedly attenuates or completely blocks, reversibly, the subjective effects of intravenously administered opioids. When co-administered with morphine, on a chronic basis, naltrexone blocks the physical dependence to morphine, heroin and other opioids. Naltrexone has few, if any, intrinsic actions besides its opioid blocking properties. However it does produce some pupillary constriction by an unknown mechanism. The administration of naltrexone is not associated with the development of tolerance or dependence. In subjects physically dependent on opioids, naltrexone will precipitate withdrawal symptomatology. Clinical studies indicate that 50 mg of naltrexone hydrochloride will block the pharmacologic effects of 25 mg of intravenously administered heroin for periods as long as 24 hours. Other data suggest that doubling the dose of naltrexone hydrochloride provides blockade for 48 hours, and tripling the dose of naltrexone hydrochloride provides blockade for about 72 hours. Naltrexone blocks the effect of opioids by competitive binding (i.e., analogous to competitive inhibition of enzymes) at opioid receptors. This makes the blockade produced potentially surmountable, but overcoming full naltrexone blockade by administration of very high doses of opiates has resulted in excessive symptoms of histamine release in experimental subjects.
The mechanism of action of naltrexone in alcoholism is not understood, however, involvement of the endogenous opioid system is suggested by preclinical data. Naltrexone, an opioid receptor antagonist, competitively binds to such receptors and may block the effects of endogenous opioids. Opioids antagonists have been shown to reduce alcohol consumption by animals, and naltrexone has been shown to reduce alcohol consumption in clinical studies. Naltrexone is not aversive therapy and does not cause a disulfiram - like reaction either as a result of opiate use or ethanol ingestion.
PHARMACOKINETICS :
Naltrexone is a pure opioid receptor antagonist. Although well absorbed orally, naltrexone is subject to significant first pass metabolism with oral bioavailability estimates ranging from 5 % to 40 %. The activity of naltrexone is believed to be due to both parent and the 6-ß-naltrexol metabolite. Both parent drug and metabolites are excreted primarily by the kidney (53 % to 79 % of the dose), however, urinary excretion of unchanged naltrexone accounts for less than 2 % of an oral dose and foecal excretion is a minor elimination pathway. The mean elimination half-life (T1/2) values for naltrexone and 6-ß-naltrexol are 4 hours and 13 hours, respectively. Naltrexone and 6-ß-naltrexol are dose proportional in terms of AUC and Cmax over the range of 50 to 200 mg and do not accumulate after 100 mg daily doses.
Absorption :
Following oral administration, naltrexone undergoes rapid and nearly complete absorption with approximately 96 % of the dose absorbed from the gastrointestinal tract. Peak plasma levels of both naltrexone and 6-ß-naltrexol occur within one hour of dosing.
Distribution :
The volume of distribution for naltrexone following intravenous administration is estimated to be 1350 litres. In vitro tests with human plasma show naltrexone to be 21 % bound to plasma proteins over the therapeutic dose range.
Metabolism : The systemic clearance (after intravenous administration) of naltrexone is ~3.5 l/min, which exceeds liver blood flow (~1.2 l/min). This suggests both that naltrexone is a highly extracted drug (> 98 % metabolized) and that extra-hepatic sites of drug metabolism exist. The major metabolite of naltrexone is 6-ß-naltrexol. Two other minor metabolites are 2-hydroxy-3-methoxy-6-ß-naltrexol and 2-hydroxy-3-methyl-naltrexone. Naltrexone and its metabolites are also conjugated to form additional metabolic products.
Elimination : The renal clearance for naltrexone ranges from 30 to 127 ml/min and suggests that renal elimination is primarily by glomerular filtration. In comparison, the renal clearance for 6-ß-naltrexol ranges from 230 to 369 ml/min, suggesting an additional renal tubular secretory mechanism. The urinary excretion of unchanged naltrexone accounts for less than 2 % of an oral dose; urinary excretion of unchanged and conjugated 6-ß-naltrexol accounts for 43 % of an oral dose. The pharmacokinetic profile of naltrexone suggests that naltrexone and its metabolites may undergo enterohepatic recycling.
Hepatic and Renal Impairment : Naltrexone appears to have extra-hepatic sites of drug metabolism and its major metabolite undergoes active tubular secretion. Adequate studies of naltrexone in patients with severe hepatic or renal impairment have not been conducted.
INDICATIONS :
Naltrexone hydrochloride tablets are indicated :
In the treatment of alcohol dependence and for the blockade of the effects of exogenously administered opioids. Naltrexone hydrochloride tablets have not been shown to provide any therapeutic benefit except as part of an appropriate plan of management for the addictions.
Administration :
For oral use.
IF THERE IS ANY QUESTION OF OCCULT OPIOID DEPENDENCE, PERFORM A NALOXONE CHALLENGE TEST AND DO NOT INITIATE NALTREXONE THERAPY UNTIL THE NALOXONE CHALLENGE IS NEGATIVE.
Dosage :
Treatment of Alcoholism :
A dose of 50 mg once daily is recommended for most patients. A patient is a candidate for treatment with naltrexone if :
- the patient is willing to take a medicine to help with alcohol dependence
- the patient is opioid free for 7 to 10 days
- the patient does not have severe or active liver or kidney problems (Typical guidelines suggest liver function tests no greater than 3 times the upper limits of normal, and bilirubin normal.)
- the patient is not allergic to naltrexone, and no other contraindications are present
Naltrexone should be considered as only one of many factors determining the success of treatment of alcoholism. Factors associated with a good outcome in the clinical trials with naltrexone were the type, intensity, and duration of treatment; appropriate management of comorbid conditions; use of community-based support groups; and good medication compliance. To achieve the best possible treatment outcome, appropriate compliance-enhancing techniques should be implemented for all components of the treatment program, especially medication compliance. The duration of treatment should not exceed 3 months.
Treatment of Opioid Dependence :
Initiate treatment with naltrexone using the following guidelines :
1.Treatment should not be attempted unless the patient has remained opioid-free for at least 7 to 10 days. Self-reporting of abstinence from opioids in opioid addicts should be verified by analysis of the patients urine for absence of opioids. The patient should not be manifesting withdrawal signs or reporting withdrawal symptoms.
2.If there is any question of occult opioid dependence, perform a naloxone challenge test. If signs of opioid withdrawal are still observed following naloxone challenge, treatment with naltrexone should not be attempted. The naloxone challenge can be repeated in 24 hours.
3.Treatment should be initiated carefully, with an initial dose of 25 mg of naltrexone hydrochloride. If no withdrawal signs occur, the patient may be started on 50 mg a day thereafter.
Naloxone Challenge Test :
The naloxone challenge test should not be performed in a patient showing clinical signs or symptoms of opioid withdrawal, or in a patient whose urine contains opioids. The naloxone challenge test may be administered by either the intravenous or subcutaneous routes.
Intravenous :
Inject 0.2 mg naloxone. Observe for 30 seconds for signs or symptoms of withdrawal. If no evidence of withdrawal, inject 0.6 mg of naloxone. Observe for an additional 20 minutes.
Subcutaneous :
Administer 0.8 mg naloxone. Observe for 20 minutes for signs or symptoms of withdrawal.
Note :
Individual patients, especially those with opioid dependence, may respond to lower doses of naloxone. In some cases, 0.1 mg IV naloxone has produced a diagnostic response.
Interpretation of the Challenge :
Monitor vital signs and observe the patient for signs and symptoms of opioid withdrawal. These may include but are not limited to : nausea, vomiting, dysphoria, yawning, sweating, tearing, rhinorrhea, stuffy nose, craving for opioids, poor appetite, abdominal cramps, sense of fear, skin erythema, disrupted sleep patterns, fidgeting, uneasiness, poor ability to focus, mental lapses, muscle aches or cramps, pupillary dilation, piloerection, fever, changes in blood pressure, pulse or temperature, anxiety, depression, irritability, backache, bone or joint pains, tremors, sensations of skin crawling, or fasciculations. If signs or symptoms of withdrawal appear, the test is positive and no additional naloxone should be administered.
Warning :
If the test is positive, do NOT initiate naltrexone therapy. Repeat the challenge in 24 hours. If the test is negative, naltrexone therapy may be started if no other contraindications are present. If there is any doubt about the result of the test, hold naltrexone and repeat the challenge in 24 hours.
Alternative Dosing Schedules :
Once the patient has been started on naltrexone hydrochloride, 50 mg every 24 hours will produce adequate clinical blockade of the actions of parenterally administered opioids (i.e., this dose will block the effects of a 25 mg intravenous heroin challenge). A flexible approach to a dosing regimen may need to be employed in cases of supervised administration. Thus, patients may receive 50 mg of naltrexone hydrochloride every weekday with a 100 mg dose on saturday, 100 mg every other day, or 150 mg every third day. The degree of blockade produced by naltrexone may be reduced by these extended dosing intervals. There may be a higher risk of hepatocellular injury with single doses above 50 mg, and use of higher doses and extended dosing intervals should balance the possible risks against the probable benefits.
Patient Compliance :
Naltrexone should be considered as only one of many factors determining the success of treatment. To achieve the best possible treatment outcome, appropriate compliance-enhancing techniques should be implemented for all components of the treatment program, including medication compliance.
PRECAUTIONS :
General :
When Reversal of Naltrexone Blockade is Required :
In an emergency situation in patients receiving fully blocking doses of naltrexone, a suggested plan of management is regional analgesia, conscious sedation with a benzodiazepine, use of non-opioid analgesics or general anaesthesia. In a situation requiring opioid analgesia, the amount of opioid required may be greater than usual, and the resulting respiratory depression may be deeper and more prolonged. A rapidly acting opioid analgesic which minimizes the duration of respiratory depression is preferred. The amount of analgesic administered should be titrated to the needs of the patient. Non-receptor mediated actions may occur and should be expected (e.g., facial swelling, itching, generalized erythema, or bronchoconstriction) presumably due to histamine release. Irrespective of the drug chosen to reverse naltrexone blockade, the patient should be monitored closely by appropriately trained personnel in a setting equipped and staffed for cardiopulmonary resuscitation.
Accidentally Precipitated Withdrawal :
Severe opioid withdrawal syndromes precipitated by the accidental ingestion of naltrexone have been reported in opioid-dependent individuals. Symptoms of withdrawal have usually appeared within five minutes of ingestion of naltrexone and have lasted for up to 48 hours. Mental status changes including confusion, somnolence and visual hallucinations have occurred. Significant fluid losses from vomiting and diarrhoea have required intravenous fluid administration. In all cases patients were closely monitored and therapy with non-opioid medications was tailored to meet individual requirements. Use of naltrexone does not eliminate or diminish withdrawal symptoms. If naltrexone is initiated early in the abstinence process, it will not preclude the patients experience of the full range of signs and symptoms that would be experienced if naltrexone had not been started. Numerous adverse events are known to be associated with withdrawal.
Special Risk Patients :
Renal Impairment :
Naltrexone and its primary metabolite are excreted primarily in the urine, and caution is recommended in administering the drug to patients with renal impairment.
Hepatic Impairment :
Caution should be exercised when naltrexone hydrochloride is administered to patients with liver disease. An increase in naltrexone AUC of approximately 5- and 10-fold in patients with compensated and decompensated liver cirrhosis, respectively, compared with subjects with normal liver function has been reported. These data also suggest that alterations in naltrexone bioavailability are related to liver disease severity.
Suicide :
The risk of suicide is known to be increased in patients with substance abuse with or without concomitant depression. This risk is not abated by treatment with naltrexone.
Pregnancy : Pregnancy Category C.
Naltrexone has been shown to increase the incidence of early foetal loss when given to rats at doses ≥ 30 mg/kg/day (180 mg/m2/day; 5 times the recommended therapeutic dose, based on body surface area) and to rabbits at oral doses ≥ 60 mg/kg/day (720 mg/m2/day; 18 times the recommended therapeutic dose, based on body surface area). There was no evidence of teratogenicity when naltrexone was administered orally to rats and rabbits during the period of major organogenesis at doses up to 200 mg/kg/day (32 and 65 times the recommended therapeutic dose, respectively, based on body surface area). Rats do not form appreciable quantities of the major human metabolite, 6-ß-naltrexol; therefore, the potential reproductive toxicity of the metabolite in rats is not known. There are no adequate and well-controlled studies in pregnant women. Naltrexone should be used during pregnancy only if the potential benefit justifies the potential risk to the foetus.
Labor and Delivery :
Whether or not naltrexone affects the duration of labor and delivery is unknown.
Nursing Mothers :
In animal studies, naltrexone and 6-ß-naltrexol were excreted in the milk of lactating rats dosed orally with naltrexone. Whether or not naltrexone is excreted in human milk is unknown. Because many drugs are excreted in human milk, caution should be exercised when naltrexone is administered to a nursing woman.
Paediatric Use :
The safe use of naltrexone in paediatric patients younger than 18 years old has not been established.
EFFECTS ON ABILITY TO DRIVE AND USE MACHINES :
Naltrexone may impair the mental and/or physical abilities required for performance of potentially hazardous tasks such as driving a car or operating machinery.
INTERACTIONS AND INCOMPATIBILITIES :
Studies to evaluate possible interactions between naltrexone and drugs other than opiates have not been performed. Consequently, caution is advised if the concomitant administration of naltrexone and other drugs is required. The safety and efficacy of concomitant use of naltrexone and disulfiram is unknown, and the concomitant use of two potentially hepatotoxic medications is not ordinarily recommended unless the probable benefits outweigh the known risks. Lethargy and somnolence have been reported following doses of naltrexone and thioridazine. Patients taking naltrexone may not benefit from opioid containing medicines, such as cough and cold preparations, antidiarrhoeal preparations, and opioid analgesics. In an emergency situation when opioid analgesia must be administered to a patient receiving naltrexone, the amount of opioid required may be greater than usual, and the resulting respiratory depression may be deeper and more prolonged.
SIDE EFFECTS :
The following adverse reactions have been reported before and during naltrexone medication :
an incidence of more than 10 % in detoxified opioid abusers : difficulty sleeping, anxiety, nervousness, abdominal pain/cramps, nausea and/or vomiting, low energy, joint and muscle pain, and headache;
an incidence of less than 10 % : loss of appetite, diarrhoea, constipation, increased thirst, increased energy, feeling down, irritability, dizziness, skin rash, delayed ejaculation, decreased potency, chills, chest pain, increased sweating and increased lacrimation. Occasional liver function abnormalities have also been reported.
ANY ABUSE AND DEPENDANCE :
Naltrexone is a pure opioid antagonist. It does not lead to physical or psychological dependence. Tolerance to the opioid antagonist effect is not known to occur.
OVERDOSAGE :
There is limited clinical experience with naltrexone overdosage in humans. In one study, subjects who received 800 mg daily naltrexone hydrochloride for up to one week showed no evidence of toxicity. In the mouse, rat and guinea pig, the oral LD50s were 1,100 to 1,550 mg/kg; 1,450 mg/kg; and 1,490 mg/kg; respectively. High doses of naltrexone hydrochloride (generally ≥ 1,000 mg/kg) produced salivation, depression/reduced activity, tremors, and convulsions. Mortalities in animals due to high-dose naltrexone administration usually were due to clonic-tonic convulsions and/or respiratory failure.
TREATMENT OF OVERDOSAGE :
In view of the lack of actual experience in the treatment of naltrexone hydrochloride overdose, patients should be treated symptomatically in a closely supervised environment.
STORAGE :
Store below 30°C, protected from moisture and light.
Do not refrigerate.
SHELF LIFE :
24 months from the date of manufacture.
PRESENTATION :
Naltrexone Hydrochloride Tablets contains 50 mg of Naltrexone HCI USP.
3 Strips of 10 Tablets per Box.
Disclaimer : For the use of a Registered Medical Practitioner or a Hospital or a Institution only. Also it is not intended to be used by healthcare professionals or patients for the purpose of prescribing or administering these products. Questions regarding the complete and current content of product labeling / specification / presentation should be directed to SGPharma.

 Cardiovascular
Cardiovascular