2.5 mg
For the use of a Registered Medical Practitioner or a Hospital or a Institution only.
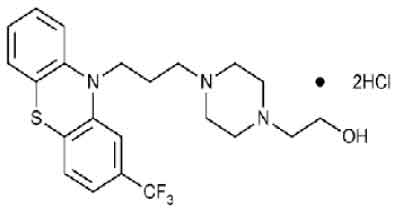
FLUPHENAZINE HYDROCHLORIDE TABLETS (Fluphenazine Hydrochloride) is a phenothiazine with general properties similar to those of chlorpromazine. It has a piperazine side - chain. Chemically, Fluphenazine Hydrochloride is 4-[3-[2-(Trifluoromethyl)phenothiazin-10-yl]propyl]-1-piperazineethanol dihydrochloride. The molecular formula is C22H26F3N3OS.2HCl and molecular weight is 510.44
STRUCTURAL FORMULA :
Its structural formula is :
FLUPHENAZINE HYDROCHLORIDE TABLETS is blue coloured circular film coated tablet.
COMPOSITION :
Each film coated tablet contains :
Fluphenazine Hydrochloride USP 2.5 mg
Excipients q.s.
Colours : Titanium Dioxide USP, Brilliant Blue.
ACTIONS :
Fluphenazine HCl is a highly potent antipsychotic agent with activity at all levels of the central nervous system as well as on multiple organ systems. The basic effects of fluphenazine hydrochloride appear to be no different from those of fluphenazine decanoate. The only exception is that the esterification of fluphenazine with decanoic acid markedly prolongs the drug’s duration of effect without reducing its activity. Like all phenothiazine derivatives, fluphenazine hydrochloride appears to act on the hypothalamus, depressing various components of the mesodiencephalic activating system which is involved in the control of basal metabolism and body temperature, wakefulness, vasomotor tone, emesis, and hormonal balance. In addition, the phenothiazines exert a peripheral autonomic affect to varying degrees. However, the site and mode of action of the phenothiazines have not been completely elucidated.
PHARMACOKINETICS :
As with all antipsychotic medications, fluphenazine is characterised by inter-individual variability in pharmacokinetics, most marked with the oral preparations. Fluphenazine is extensively metabolized, undergoing “first pass” metabolism by the liver, and is excreted in both the urine and faeces. The degree of antipsychotic activity of the metabolites is still unknown. Fluphenazine is highly protein-bound (greater than 90 %) in plasma. With oral or I.M. fluphenazine HCl, peak plasma/serum levels are attained within a few hours. The serum half life of intramuscular and oral fluphenazine HCl is approximately 15 hours. Phenothiazines cross the blood-brain barrier, cross the placenta easily, and cannot be removed by dialysis. It is not known whether fluphenazine is present in breast milk. However, other phenothiazines have been shown to be excreted in human breast milk.
INDICATIONS :
FLUPHENAZINE HYDROCHLORIDE TABLETS are indicated in the management of manifestations of psychotic disorders including schizophrenia and manic psychosis. FLUPHENAZINE HYDROCHLORIDE TABLETS has not been shown effective in the management of behavioral complications in patients with mental retardation.
Administration :
FLUPHENAZINE HYDROCHLORIDE TABLETS is for oral administration.
Dosage :
Depending on severity and duration of symptoms, total daily oral dosage for adult psychotic patients may range initially from 2.5 mg to 10 mg and should be divided and given at 6 to 8 hour intervals. The smallest amount that will produce the desired results must be carefully determined for each individual, since optimal dosage levels of this potent drug vary from patient to patient. In general, the oral dose has been found to be approximately two to three times the parenteral dose of fluphenazine. Treatment is best instituted with a low initial dosage, which may be increased, if necessary, until the desired clinical effects are achieved. Therapeutic effect is often achieved with doses under 20 mg daily. Patients remaining severely disturbed or inadequately controlled may require upward titration of dosage. Daily doses up to 40 mg may be necessary; controlled clinical studies have not been performed to demonstrate safety of prolonged administration of such doses. When symptoms are controlled, dosage can generally be reduced gradually to daily maintenance doses of 1 mg or 5 mg, often given as a single daily dose. Continued treatment is needed to achieve maximum therapeutic benefits; further adjustments in dosage may be necessary during the course of therapy to meet the patient’s requirements (See fluphenazine decanoate package insert). For psychotic patients who have been stabilized on a fixed daily dosage of orally administered fluphenazine hydrochloride dosage forms, conversion to the long-lasting injectable fluphenazine decanoate injection may be indicated. For geriatric patients, the suggested starting dose is 1 mg to 2.5 mg daily, adjusted according to the response of the patient.
CONTRAINDICATIONS :
Phenothiazines are contraindicated in patients with suspected or established subcortical brain damage, in patients receiving large doses of hypnotics, and in comatose or severely depressed states. The presence of blood dyscrasia or liver damage precludes the use of fluphenazine hydrochloride. FLUPHENAZINE HYDROCHLORIDE TABLETS are contraindicated in patients who have shown hypersensitivity to fluphenazine; cross-sensitivity to phenothiazine derivatives may occur. FLUPHENAZINE HYDROCHLORIDE TABLETS contains lactose which is contra-indicated in patients with galactosaemia, the glucose-galactose malabsorption syndrome, or lactase deficiency.
WARNINGS :
Increased Mortality in Elderly Patients with Dementia - Related Psychosis :
Elderly patients with dementia – related psychosis treated with antipsychotic drugs are at an increased risk of death. Fluphenazine hydrochloride is not approved for the treatment of patients with dementia – related psychosis. (See BOXED WARNING)..
Tardive Dyskinesia :
Tardive dyskinaesia, a syndrome consisting of potentially irreversible, involuntary, dyskinaetic movements may develop in patients treated with neuroleptic (antipsychotic) drugs. Although the prevalence of the syndrome appears to be highest among the elderly, especially elderly women, it is impossible to rely upon prevalence estimates to predict, at the inception of neuroleptic treatment, which patients are likely to develop the syndrome. Whether neuroleptic drug products differ in their potential to cause tardive dyskinaesia is unknown. Both the risk of developing the syndrome and the likelihood that it will become irreversible are believed to increase as the duration of treatment and total cumulative dose of neuroleptic drugs administered to the patient increase. However, the syndrome can develop, although much less commonly, after relatively brief treatment periods at low doses. There is no known treatment for established cases of tardive dyskinaesia, although the syndrome may remit, partially or completely. If neuroleptic treatment is withdrawn. Neuroleptic treatment, itself, however, may suppress (or partially suppress) the signs and symptoms of the syndrome and thereby may possibility mask the underlying disease process. The effect that symptomatic suppression has upon the long – term course of the syndrome is unknown. Given these considerations, neuroleptics should be prescribed in a manner that is most likely to minimize the occurrence of tardive dyskinaesia. Chronic neuroleptic treatment should generally be reserved for patients who suffer from a chronic illness that,
1) is known to respond to neuroleptic drugs, and
2) for whom alternative, equally effective, but potentially less harmful treatments are not available or appropriate. In patients who do require chronic treatment, the smallest dose and the shortest duration of treatment producing a satisfactory clinical response should be sought. The need for continued treatment should be reassessed periodically.
If signs and symptoms of tardive dyskinaesia appear in a patient on neuroleptics, drug discontinuation should be considered. However, some patients may require treatment despite the presence of the syndrome.
Neuroleptic Malignant Syndrome (NMS) :
A potentially fatal symptom complex sometimes referred to as Neuroleptic Malignant Syndrome (NMS) has been reported in association with antipsychotic drugs. Clinical manifestations of NMS are hyperpyrexia, muscle rigidity, altered mental status and evidence of autonomic instability (irregular pulse or blood pressure, tachycardia, diaphoresis and cardiac dysrhythmias).
The diagnostic evaluation of patients with this syndrome is complicated. In arriving at a diagnosis, it is important to identify cases where the clinical presentation includes both serious medical illness (e.g., pneumonia, systemic infection etc.) and untreated or inadequately treated extrapyramidal signs and symptoms (EPS). Other important considerations in the differential diagnosis include central anticholinergic toxicity, heat stroke, drug fever and primary central nervous system (CNS) pathology.
The management of NMS should include :
1) Immediate discontinuation of antipsychotic drugs and other drugs not essential to concurrent therapy,
2) Intensive symptomatic treatment and medical monitoring and
3) Treatment of any concomitant serious medical problems for which specific treatments are available. There is no general agreement about specific pharmacological treatment regimens for uncomplicated NMS.
If a patient requires antipsychotic drug treatment after recovery from NMS, the potential reintroduction of drug therapy should be carefully considered. The patient should be carefully monitored. Since recurrences of NMS have been reported. The use of this drug may impair the mental and physical abilities required for driving a car or operating heavy machinery. Potentiation of the effects of alcohol may occur with the use of this drug. Since there is no adequate experience in children who have received this drug, safety and efficacy in children have not been established.
PRECAUTION :
General :
Because of the possibility of cross-sensitivity, FLUPHENAZINE HYDROCHLORIDE TABLETS should be used cautiously in patients who have developed cholestatic jaundice, dermatoses, or other allergic reactions to phenothiazine derivatives. Psychotic patients on large doses of a phenothiazine drug who are undergoing surgery should be watched carefully for possible hypotensive phenomena. Moreover, it should be remembered that reduced amounts of anaesthetics or central nervous system depressants may be necessary. The effects of atropine may be potentiated in some patients receiving fluphenazine because of added anticholinergic effects. FLUPHENAZINE HYDROCHLORIDE TABLETS should be used cautiously in patients exposed to extreme heat or phosphorous insecticides; in patients with a history of convulsive disorders, since grand mal convulsions have been known to occur; and in patients with special medical disorders, such as mitral insufficiency or other cardiovascular diseases and pheochromocytoma. The possibility of liver damage, pigmentary retinopathy, lenticular and corneal deposits, and development of irreversible dyskinaesia should be remembered when patients are on prolonged therapy.
Neuroleptic drugs elevate prolactin levels; the elevation persists during chronic administration. Tissue culture experiments indicate that approximately one-third of human breast cancers are prolactin dependent in vitro, a factor of potential importance if the prescription of these drugs is contemplated in a patient with a previously detected breast cancer. Although disturbances such as galactorrhoea, amenorrhoea, gynaecomastia, and impotence have been reported, the clinical significance of elevated serum prolactin levels is unknown for most patients. An increase in mammary neoplasms has been found in rodents after chronic administration of neuroleptic drugs. Neither clinical studies nor epidemiologic studies conducted to date, however, have shown an association between chronic administration of these drugs and mammary tumorigenesis; the available evidence is considered too limited to be conclusive at this time.
Pregnancy : FDA pregnancy categories are not included in product labeling presently.
Phenothiazines are not recommended for use during pregnancy. Phenothiazines cross placenta. Although adequate and well controlled studies in humans have not been done, there have been reports of prolonged jaundice, hyporeflexia or hyperreflexia and extrapyramidal effects in the neonates of mothers who received phenothiazines during pregnancy. Withdrawal effects, including severe rhinorrhoea, vomiting, respiratory distress and extrapyramidal effects have been reported in neonates following in utero exposure to fluphenazine throughout gestation.
Breast feeding :
Phenothiazines are distributed into breast milk and may cause drowsiness or movement disorders in the nursing infant. Breast feeding while receiving phenothiazines is not recommended.
Paediatric use :
Children are prone to develop neuromuscular or extrapyramidal reactions, especially dystonias and should be closely monitored while receiving therapeutic doses of phenothiazines. Children with acute illnesses, such as chickenpox, central nervous system (CNS) infections, measles, gastroenteritis and dehydration are especially at risk.
Geriatric use :
Geriatric patients tend to develop higher plasma concentrations of Phenothiazines. Therefore, these patients usually require lower initial dosage and a more gradual titration of dosage. Elderly patients appear to be more prone to orthostatic hypotension and exhibit an increased sensitivity to the anticholinergic and sedative effects of phenothiazines. In addition, they are more prone to develop extrapyramidal side effects such as tardive dyskinaesia and parkinsonism. It has been suggested that elderly patients receive half the usual adult dose. Patients with organic mental disorders or acute confusional states should initially receive one third to one half the usual adult dose, with the dose being increased no more frequently than every 2 or 3 days, preferably at intervals of 7 to 10 days, if possible. After clinical improvement occurs, periodic attempts should be made to discontinue medication.
INTERACTIONS :
The possibility should be borne in mind that phenothiazines may :
1. Increase the central nervous system depression produced by drugs such as alcohol, hypnotics, sedatives or strong analgesics.
2. Antazonize the action of adrenaline and other sympathomimetic agents and reverse the blood pressure-lowering effects of adrenergic-blocking agents such as guanethidine and clonidine.
3. Impair :
The anti-Parkinsonian effect of L-Dopa;
The effect of anti-convulsants;
The metabolism of tricyclic antidepressants;
The control of diabetes.
4. Increase the effect of anticoagulants.
5. Interact with lithium in terms predisposing to adverse reactions. Interact with anticholinergic drugs with possible enhancement of its anticholinergic effects. For example, an anti-Parkinson drug, such as benserazide, used to manage extrapyramidal side effects, may exacerbate the antimuscarinic effects of fluphenazine.
6. Enhance the cardiac depressant effects of quinidine.
7. Enhance the absorption of corticosteroids, digoxin and neuromuscular blocking agents.
8. Give false results on pregnancy tests.
SIDE EFFECTS :
Central Nervous System :
The side effects most frequently reported with phenothiazine compounds are extrapyramidal symptoms including pseudoparkinsonism, dystonia, dyskinaesia, akathaisia, oculogyric crises, opisthotonos, and hyperreflexia. Most often these extrapyramidal symptoms are reversible; however, they may be persistent (see below). With any given phenothiazine derivative, the incidence and severity of such reactions depend more on individual patient sensitivity than on other factors, but dosage level and patient age are also determinants.
Extrapyramidal reactions may be alarming, and the patient should be forewarned and reassured. These reactions can usually be controlled by administration of antiparkinsonian drugs such as benztropine mesylate or intravenous caffeine and sodium benzoate injection, and by subsequent reduction in dosage.
Extrapyramidal Symptoms :
Dystonia
Class Effect : Symptoms of dystonia, prolonged abnormal contractions of muscle groups, may occur in susceptible individuals during the first few days of treatment. Dystonic symptoms include : spasm of the neck muscles, sometimes progressing to tightness of the throat, swallowing difficulty, difficulty breathing, and/or protrusion of the tongue. While these symptoms can occur at low doses, they occur more frequently and with greater severity with high potency and at higher doses of first generation antipsychotic drugs. An elevated risk of acute dystonia is observed in males and younger age groups.
Tardive Dyaskinesia :
See WARNINGS. The syndrome is characterized by involuntary choreoathetoid movements which variously involve the tongue, face, mouth, lips, or jaw (e.g., protrusion of the tongue, puffing of cheeks, puckering of the mouth, chewing movements), trunk and extremities. The severity of the syndrome and the degree of impairment produced vary widely. The syndrome may become clinically recognizable either during treatment, upon dosage reduction, or upon withdrawal of treatment. Early detection of tardive dyskinaesia is important. To increase the likelihood of detecting the syndrome at the earliest possible time, the dosage of neuroleptic drug should be reduced periodically (if clinically possible) and the patient observed for signs of the disorder. This maneuver is critical, since neuroleptic drugs may mask the signs of the syndrome.
Other CNS Effects :
Occurrences of neuroleptic malignant syndrome (NMS) have been reported in patients on neuroleptic therapy (see WARNINGS : Neuroleptic Malignant Syndrome); leukocytosis, elevated CPK, liver function abnormalities, and acute renal failure may also occur with NMS. Drowsiness or lethargy, if they occur, may necessitate a reduction in dosage; the induction of a catatonic-like state has been known to occur with dosages of fluphenazine far in excess of the recommended amounts. As with other phenothiazine compounds, reactivation or aggravation of psychotic processes may be encountered. Phenothiazine derivatives have been known to cause, in some patients, restlessness, excitement, or bizarre dreams.
Autonomic Nervous System :
Hypertension and fluctuations in blood pressure have been reported with fluphenazine hydrochloride. Hypotension has rarely presented a problem with fluphenazine. However, patients with pheochromocytoma, cerebral vascular or renal insufficiency, or a severe cardiac reserve deficiency such as mitral insufficiency appear to be particularly prone to hypotensive reactions with phenothiazine compounds, and should therefore be observed closely when the drug is administered. If severe hypotension should occur, supportive measures including the use of intravenous vasopressor drugs should be instituted immediately. Norepinephrine bitartrate injection is the most suitable drug for this purpose; epinephrine should not be used since phenothiazine derivatives have been found to reverse its action, resulting in a further lowering of blood pressure. Autonomic reactions including nausea and loss of appetite, salivation, polyuria, perspiration, dry mouth, headache, and constipation may occur. Autonomic effects can usually be controlled by reducing or temporarily discontinuing dosage. In some patients, phenothiazine derivatives have caused blurred vision, glaucoma, bladder paralysis, faecal impaction, paralytic ileus, tachycardia, or nasal congestion.
Metabolic and Endocrine :
Weight change, peripheral oedema, abnormal lactation, gynaecomastia, menstrual irregularities, false results on pregnancy tests, impotency in men and increased libido in women have all been known to occur in some patients on phenothiazine therapy.
Allergic Reactions :
Skin disorders such as itching, erythema, urticaria, seborrhoea, photosensitivity, eczema and even exfoliative dermatitis have been reported with phenothiazine derivatives. The possibility of anaphylactoid reactions occurring in some patients should be borne in mind.
Haematologic :
Routine blood counts are advisable during therapy since blood dyscrasias including leukopaenia, agranulocytosis, thrombocytopaenic or nonthrombocytopaenic purpura, eosinophilia, and pancytopaenia have been observed with phenothiazine derivatives. Furthermore, if any soreness of the mouth, gums, or throat, or any symptoms of upper respiratory infection occur and confirmatory leuokocyte count indicates cellular depression, therapy should be discontinued and other appropriate measures instituted immediately.
Hepatic :
Liver damage as manifested by cholestatic jaundice may be encountered, particularly during the first months of therapy; treatment should be discontinued if this occurs. An increase in cephalin flocculation, sometimes accompanied by alterations in other liver function tests, has been reported in patients receiving fluphenazine hydrochloride who have had no clinical evidence of liver damage.
Others :
Sudden, unexpected and unexplained deaths have been reported in hospitalized psychotic patients receiving phenothiazines. Previous brain damage or seizures may be predisposing factors; high doses should be avoided in known seizure patients. Several patients have shown sudden flare-ups of psychotic behavior patterns shortly before death. Autopsy findings have usually revealed acute fulminating pneumonia or pneumonitis, aspiration of gastric contents, or intramyocardial lesions. Although this is not a general feature of fluphenazine, potentiation of central nervous system depressants (opiates, analgesics, antihistamines, barbiturates, alcohol) may occur. The following adverse reactions have also occurred with phenothiazine derivatives : systemic lupus erythematosus-like syndrome, hypotension severe enough to cause fatal cardiac arrest, altered electrocardiographic and electroencephalographic tracings, altered cerebrospinal fluid proteins, cerebral oedema, asthma, laryngeal oedema, and angioneurotic oedema; with long-term use–skin pigmentation, and lenticular and corneal opacities.
INFORMATION FOR PATIENTS :
Given the likelihood that some patients exposed chronically to neuroleptics will develop tardive dyskinesia, it is advised that all patients in whom chronic use is contemplated be given, if possible, full information about this risk. The decision to inform patients and/or their guardians must obviously take into account the clinical circumstances and the competency of the patient to understand the information provided.
OVERDOSAGE :
In general, the symptoms of overdose are extensions of known pharmacologic effects and adverse reactions, the most prominent of which would be :
1. Severe extrapyramidal reactions,
2. Hypotension, or
3. Sedation.
CNS depression may progress to coma with areflexia. Restlessness, confusion and excitement may occur with early or mild intoxication.
TREATMENT OF OVERDOSAGE :
The drug should be withdrawn and the symptoms of overdose treated supportively. Up to several hours after an oral overdose, gastric lavage should be attempted, followed by activated charcoal and then cathartics. If severe hypotension should occur, supportive measures, including the use of intravenous vasopressor drugs, should be instituted immediately. Noradrenaline tartrate is the most suitable drug for this purpose; adrenaline should not be used, since phenothiazine derivatives have been found to reverse its action, resulting in a further lowering of blood pressure. In case of severe extrapyramidal reactions, antiparkinson medication should be administered, and should be continued for several weeks. Antiparkinson medication should be withdrawn gradually to avoid the mergence of rebound extrapyramidal symptoms. Limited experience indicates that phenothiazines are not dialyzable.
STORAGE :
Store below 30°C, protected from moisture and light. Do not refrigerate.
Avoid excessive heat.
SHELF LIFE :
24 months from the date of manufacture.
PRESENTATION :
FLUPHENAZINE HYDROCHLORIDE TABLETS contains Fluphenazine Hydrochloride USP 2.5 mg.
2 Strips of 10 Tablets in a carton.
Disclaimer : For the use of a Registered Medical Practitioner or a Hospital or a Institution only. Also it is not intended to be used by healthcare professionals or patients for the purpose of prescribing or administering these products. Questions regarding the complete and current content of product labeling / specification / presentation should be directed to SGPharma.

 Cardiovascular
Cardiovascular