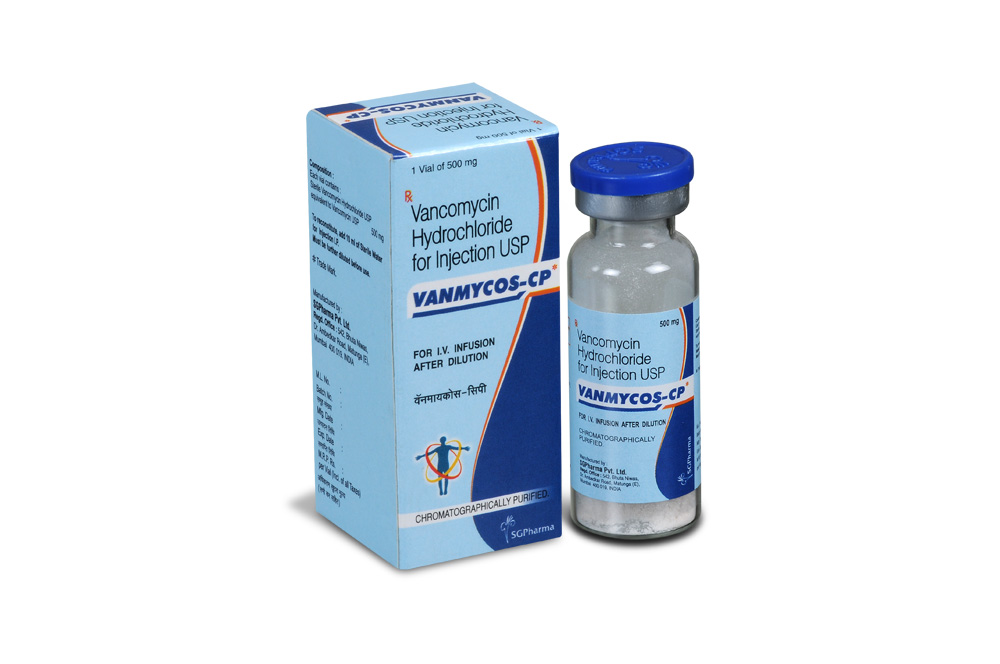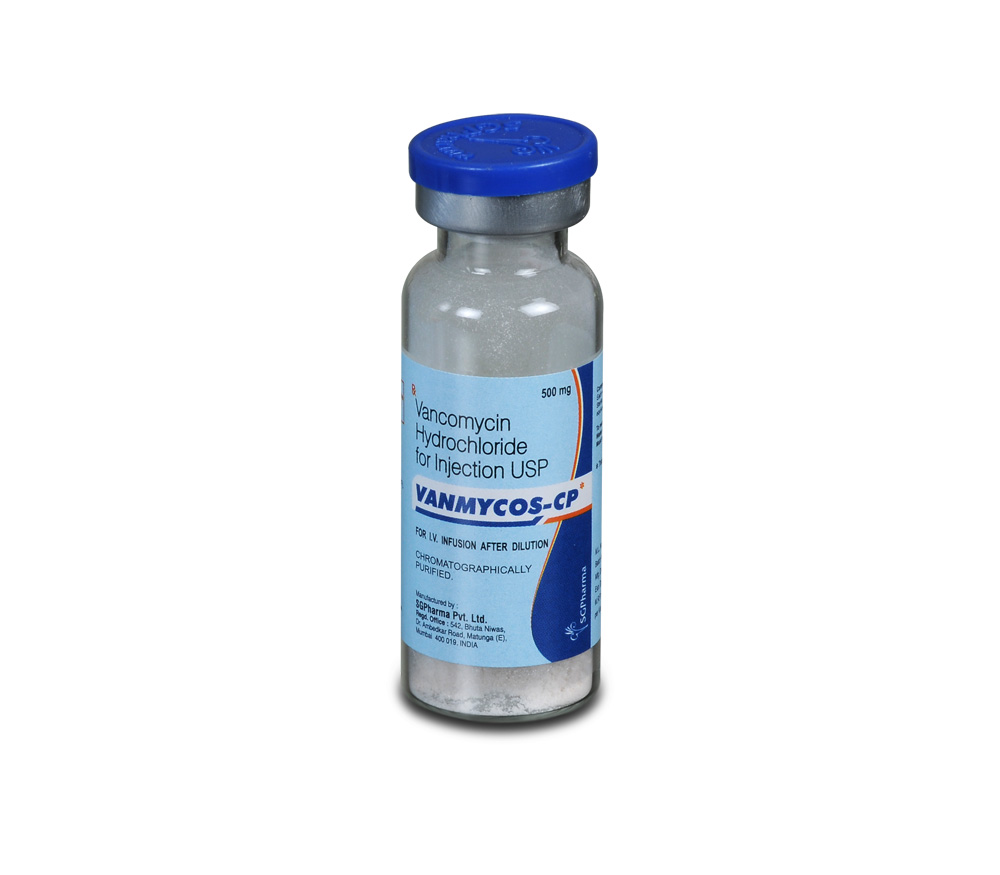
500 mg, 1 gm
For the use of a Registered Medical Practitioner or a Hospital or a Institution only.
VANCOMYCIN HYDROCHLORIDE FOR INJECTION (Vancomycin) is tricyclic glycopeptide antibiotic obtained from cultures of Nocardia orientalis. Chemically, Vancomycin Hydrochloride is (Sa)-(3S,6R,7R,22R,23S,26S,36R,38aR)-44-[[2-O-(3-Amino-2,3,6-trideoxy-3-C-methyl-α-L-lyxo-hexopyranosyl)-β-D-glucopyranosyl]oxy]-3-(carbamoylmethyl)-10,19-dichloro-2,3,4,5,6,7,23,24,25,26,36,37,38,38a-tetradecahydro-7,22,28,30,32-pentahydroxy-6-[(2R)-4-methyl-2 (methylamino)]valeramido]-2,5,24,38,39-pentaoxo-22H-8,11: 18,21-dietheno-23,36-(iminomethano) -13,16 : 31,35-dimetheno-1H,16H-[1,6,9]oxadiazacyclohexadecino[4,5-m] [10,2,16] benzoxadiazacyclotetracosine-26-carboxylic acid, monohydro-chloride. The molecular formula is C66H75Cl2N9O24.HCl and molecular weight is 1485.71.
STRUCTURAL FORMULA :
Its structural formula is :
-Structure.jpg)
VANCOMYCIN HYDROCHLORIDE FOR INJECTION is a white to light brown sterile powder filled in flint tubular vial of suitable size.
COMPOSITION :
VANCOMYCIN HYDROCHLORIDE FOR INJECTION Each vial contains :
Sterile Vancomycin Hydrochloride USP
equivalent to Vancomycin USP 500 mg
ACTIONS :
Vancomycin is a biological material, described as a tricyclic glycopeptide obtained from cultures of Nocardia orientalis (Streptomyces orientalis). Vancomycin is present as the hydrochloride salt for parenteral administration. The drug is not absorbed from the gastrointestinal tract, and an aqueous solution of the product can be administered orally in the treatment of pseudomembranous colitis. Vancomycin is a bactericidal antibiotic and appears to bind to the bacterial cell wall causing blockage of glycopeptide polymerisation. This effect produces immediate inhibition of cell wall synthesis and secondary damage to the cytoplasmic membrane. It is active against many gram positive organisms including staphylococci, group A beta haemolytic streptococci, Streptococcus pneumoniae, enterococci, corynebacterium and clostridium species. It does not demonstrate clinical efficacy against gram negative bacteria, fungi or yeasts, and hence the product literature only indicates use in severe infections caused by gram positive organisms.
MICROBIOLOGY :
The bactericidal action of Vancomycin results primarily from inhibition of cell-wall biosynthesis. In addition, Vancomycin alters bacterial-cell-membrane permeability and RNA synthesis. There is no cross-resistance between Vancomycin and other antibiotics. Vancomycin is active against staphylococci, including Staphylococcus aureus and Staphylococcus epidermidis (including heterogeneous methicillin-resistant strains); streptococci, including Streptococcus pyogenes, Streptococcus pneumoniae (including penicillin-resistant strains), Streptococcus agalactiae, the viridans group, Streptococcus bovis, and enterococci (e.g. Streptococcus faecalis); Clostridium difficile (e.g. toxigenic strains implicated in pseudomembranous enterocolitis); diphtheroids (e.g. JK corynebacterium) and Propionibacterium. Other organisms that are susceptible to Vancomycin in vitro include Listeria monocytogenes, Lactobacillus species, Actinomyces species, Clostridium species, and Bacillus species.
Vancomycin is not active in vitro against gram-negative bacilli, mycobacteria, or fungi. The combination of Vancomycin hydrochloride and an aminoglycoside acts synergistically in vitro against many strains of S. aureus, nonenterococcal group D streptococci, enterococci, and Streptococcus species (viridans group). The combination of Vancomycin and a cephalosporin may act synergistically against some strains of S. epidermidis (methicillin-resistant). The combination of Vancomycin and rifampicin acts with partial synergism against some strains of S. aureus and with synergism against S. epidermidis. Synergy testing may be helpful, the combination of Vancomycin and rifampicin may act antagonistically against some strains of S. aureus.
PHARMACOKINETICS :
Vancomycin hydrochloride is poorly absorbed after oral administration; it is given intravenously for treatment of systemic infections. Intramuscular injection is painful. In subjects with normal kidney function, multiple intravenous dosing of 1 g of Vancomycin (15 mg/kg) infused over 60 minutes produces mean plasma concentrations of approximately 63 mg/L immediately at the completion of infusion, mean plasma concentrations of approximately 23 mg/L 2 hours after infusion, and mean plasma concentrations of approximately 8 mg/L
11 hours after the end of the infusion. Multiple dosing of 500 mg infused over 30 minutes produces mean plasma concentrations of about 49 mg/L at the completion of infusion, mean plasma concentrations of about 19 mg/L 2 hours after infusion, and mean plasma concentrations of about 10 mg/L 6 hours after infusion. The plasma concentrations during multiple dosing are similar to those after a single dose. The mean elimination half-life of Vancomycin from plasma is 4-6 hours in subjects with normal renal function. In the first
24 hours, about 75 % of an administered dose of Vancomycin is excreted in urine by glomerular filtration. Mean plasma clearance is about 0.058 L/kg/hr, and mean renal clearance is about 0.048 L/kg/hr. Renal dysfunction slows excretion of Vancomycin. In anephric patients, the average half-life of elimination is 7.5 days.
The distribution coefficient is from 0.3-0.43 L/kg. There is no apparent metabolism of the medicine. About 60 % of an intraperitoneal dose of Vancomycin hydrochloride administered during peritoneal dialysis is absorbed systemically in 6 hours. Serum concentrations of about 10 mg/L are achieved by intraperitoneal injection of 30 mg/kg of Vancomycin. Vancomycin is not effectively removed by either haemodialysis or peritoneal dialysis; there have been no reports of Vancomycin clearance with haemoperfusion. Total systemic and renal clearance of Vancomycin may be reduced in the elderly. Vancomycin is approximately 55 % serum protein bound as measured by ultrafiltration at Vancomycin serum concentrations of 10-100 mg/L. After intravenous administration of Vancomycin hydrochloride, inhibitory concentrations are present in pleural, pericardial, ascitic, and synovial fluids; in urine; in peritoneal dialysis fluid; and in atrial appendage tissue. Vancomycin hydrochloride does not readily diffuse across normal meninges into the spinal fluid; but, when the meninges are inflamed, penetration into the spinal fluid occurs. Measurable serum concentrations of Vancomycin may occur in patients treated for active pseudomembranous colitis due to Clostridium difficile.
INDICATIONS :
I.V. Vancomycin hydrochloride is used in the treatment of potentially life-threatening infections caused by susceptible organisms which cannot be treated with other effective, less toxic anti-infective agents.
1.Staphylococcal infections such as endocarditis, osteomyelitis, pneumonia, septicaemia and skin and soft tissue infections caused by Staphylococcus aureus or S. epidermidis including methicillin-resistant strains.
2.Streptococcal infections ; Enterococcal endocarditis.
3.Bacterial endocarditis : Vancomycin hydrochloride is indicated for the treatment of serious or severe infections caused by susceptible strains of methicillin-resistant (beta lactam-resistant) staphylococci. It is indicated for penicillin-allergic patients, for patients who cannot receive or who have failed to respond to other medicines, including the penicillins or cephalosporins, and for infections caused by Vancomycin-susceptible organisms that are resistant to other antimicrobial medicines. Vancomycin hydrochloride is indicated for initial therapy when methicillin-resistant staphylococci are suspected, but after susceptibility data are available, therapy should be adjusted accordingly. Vancomycin hydrochloride is effective in the treatment of staphylococcal endocarditis.
4.Other Staphylococcal infections : The effectiveness of Vancomycin has been documented in other infections due to staphylococci, including septicaemia, bone infections, lower respiratory tract infections, and skin and skin structure infections. When staphylococcal infections are localised and purulent, antibiotics are used as adjuncts to appropriate surgical measures.
5.Vancomycin hydrochloride has been reported to be effective alone or in combination with an aminoglycoside for endocarditis caused by Streptococcus viridans or S. bovis. For endocarditis caused by enterococci (e.g. E. faecalis), Vancomycin hydrochloride has been reported to be effective only in combination with an aminoglycoside.
6.Vancomycin hydrochloride has been reported to be effective for the treatment of diphtheroid endocarditis. Vancomycin hydrochloride has been used successfully in combination with either rifampin, an aminoglycoside, or both in early-onset prosthetic valve endocarditis caused by S. epidermidis or diphtheroids. Specimens for bacteriologic cultures should be obtained in order to isolate and identify causative organisms and to determine their susceptibilities to Vancomycin hydrochloride.
7.The parenteral form of Vancomycin hydrochloride may be administered orally for treatment of antibiotic-associated pseudomembranous colitis produced by C. difficile. Parenteral administration of Vancomycin hydrochloride alone is of unproven benefit for this indication. Vancomycin hydrochloride is not effective by the oral route for other types of infection. Although no controlled clinical efficacy studies have been conducted, intravenous Vancomycin has been suggested by the American Dental Association as prophylaxis against bacterial endocarditis in penicillin-allergic patients who have congenital heart disease or rheumatic or other acquired valvular heart disease when these patients undergo dental procedures or surgical procedures of the upper respiratory tract.
Note : When selecting antibiotics for the prevention of bacterial endocarditis, the physician or dentist should read the full joint statement of the American Heart Association and the American Dental Association.
Administration :
For intravenous infusion and oral use only and not for intramuscular administration.
Dosage :
Patients with Normal Renal Function :
Adults :
The usual daily intravenous dose is 2 g divided either as 500 mg every six hours or 1 g every 12 hours. Each dose should be administered at no more than 10 mg/min or over a period of at least 60 minutes, whichever is longer. Other patient factors, such as age or obesity, may call for modification of the usual daily intravenous dose.
Children :
The usual intravenous dosage of Vancomycin hydrochloride, is 10 mg/kg per dose given every six hours. Each dose should be administered over a period of at least 60 minutes.
Infants and Neonates :
In neonates and young infants, the total daily intravenous dosage may be lower. In both neonates and infants, an initial dose of 15 mg/kg is suggested, followed by 10 mg/kg every 12 hours for neonates in the first week of life and every eight hours thereafter up to the age of one month. Each dose should be administered over 60 minutes. Close monitoring of serum concentrations of Vancomycin may be warranted in these patients.
Patients with Impaired Renal Function and Elderly Patients :
Dosage adjustment must be made in patients with impaired renal function. In premature infants and the elderly, greater dosage reductions than expected may be necessary because of decreased renal function. Measurement of Vancomycin serum concentrations can be determined by use of microbiologic assay, radioimmunoassay, fluorescence polarisation immunoassay, fluorescence immunoassay, or high-pressure liquid chromato-graphy. If creatinine clearance can be measured or estimated accurately, the dosage for most patients with renal impairment can be calculated using the following table. The dosage of Vancomycin hydrochloride per day in mg is about 15 times the glomerular filtration rate in mL/min :
The initial dose should be no less than 15 mg/kg, even in patients with mild to moderate renal insufficiency. The table is not valid for functionally anephric patients. For such patients, an initial dose of 15 mg/kg of body weight should be given to achieve prompt therapeutic serum concentrations. The dose required to maintain stable concentrations is 1.9 mg/kg/24h. In patients with marked renal impairment, it may be more convenient to give maintenance doses of 250 to 1,000 mg once every several days rather than administering the medicine on a daily basis. In anuria, a dose of 1,000 mg every 7-10 days has been recommended. When only the serum creatinine concentration is known, the following formula (based on sex, weight and age of the patient) may be used to calculate creatinine clearance. Calculated creatinine.
CONTRAINDICATIONS :
Vancomycin is contraindicated in patients showing hypersensitivity to Vancomycin or any component of the formulation. It should be avoided in patients who have an underlying hearing loss.
WARNINGS :
Rapid bolus administration (e.g. over several minutes) may be associated with exaggerated hypotension, including shock, and, rarely, cardiac arrest. Vancomycin should be infused in a dilute solution over a period of not less than 60 minutes to avoid rapid infusion-related reactions. Stopping the infusion usually results in a prompt cessation of these reactions. Some patients with inflammatory disorders of the intestinal mucosa may have significant systemic absorption of oral Vancomycin and, therefore, may be at risk for the development of adverse reactions associated with the parenteral administration of Vancomycin. The risk is greater in patients with renal impairment. It should be noted that the total systemic and renal clearances of Vancomycin are reduced in the elderly. Due to its potential ototoxicity and nephrotoxicity, Vancomycin should be used with care in patients with renal insufficiency and the dose should be reduced according to the degree of renal impairment. The risk of toxicity is appreciably increased by high blood concentrations or prolonged therapy. Blood levels should be monitored and renal function tests should be performed regularly.
Vancomycin should also be avoided in patients with previous hearing loss. If it is used in such patients, the dose should be regulated, if possible, by periodic determination of the drug level in the blood. Deafness may be preceded by tinnitus. The elderly are more susceptible to auditory damage. Experience with other antibiotics suggests that deafness may be progressive despite cessation of treatment. Redman (or Redneck) syndrome : This is characterized by a sudden and profound fall in blood pressure with or without maculopapular rash over the face, neck, upper chest and extremities. This reaction appears to be partially mediated through a histaminergic response stimulated due to rapid I.V. infusion. Therefore I.V. Vancomycin should be administered as a slow intravenous infusion.
PRECAUTIONS :
Clinically significant serum concentrations have been reported in some patients being treated for active C. difficile-induced pseudomembranous colitis after multiple oral doses of Vancomycin. Therefore, monitoring of serum concentrations may be appropriate in these patients. Patients with borderline renal function and individuals over the age of 60 should be given serial tests of auditory function and of Vancomycin blood levels. All patients receiving the drug should have periodic haematological studies, urine analysis and renal function tests. Vancomycin is very irritating to tissue, and causes injection site necrosis when injected intramuscularly; it must be infused intravenously. Injection site pain and thrombophlebitis occur in many patients receiving Vancomycin and are occasionally severe.
The frequency and severity of thrombophlebitis can be minimised by administering the drug slowly as a dilute solution (2.5 to 5.0 g/L) and by rotating the sites of infusion. Prolonged use of Vancomycin may result in the overgrowth of non-susceptible organisms. Careful observation of the patient is essential.If superinfection occurs during therapy, appropriate measures should be taken. In rare instances, there have been reports of pseudomembranous colitis, due to C. difficile, developing in patients who received intravenous Vancomycin.
Patients taking oral Vancomycin should be warned of its offensive taste.
Pregnancy : Category C
Use in pregnancy only if clearly indicated.
Lactation :
Vancomycin is excreted in breast milk. Caution should be exercised when administering drug to a nursing women. Because of the potential for adverse events, a decision has to be taken whether to discontinue nursing or to discontinue the drug, taking into account the importance of the drug to the mother.
Paediatric use :
Vancomycin should be used with caution in premature neonates and young infants because of the renal immaturity of these patients and the potential for increased serum concentrations of the drug. Close monitoring of serum Vancomycin concentrations may be warranted in neonates and young infants concomitant administration of Vancomycin and anaesthetic agents in children has been associated with erythema and histamine - like flushing.
Elderly use :
Because geriatric adults may have decreasing glomerular filtration with increasing age, increased serum Vancomycin concentrations may occur if dosage is not adjusted in these patients. Vancomycin dosage in geriatric patients should be adjusted based on the degree of renal impairment.
INTERACTIONS :
Concurrent administration of Vancomycin and anaesthetic agents has been associated with erythema, histamine like flushing and anaphylactoid reactions. Concurrent administration with other neurotoxic or nephrotoxic drugs, e.g. amphotericin B, streptomycin, neomycin, gentamicin, kanamycin, amikacin, tobramycin, viomycin, bacitracin, polymyxin B, colistin and cisplatin requires careful monitoring. Diuretics such as ethacrynic acid and frusemide may aggravate ototoxicity. Cholestyramine has been shown to bind Vancomycin in vitro. Therefore, if oral Vancomycin is used with cholestyramine, the two drugs should be administered several hours apart.
SIDE EFFECTS :
Auditory and Vestibular :
Sensorineural deafness which may be accompanied by tinnitus has occurred but the incidence is low. Permanent deafness is more likely to occur in patients with compromised auditory or renal function but reversible deafness has been reported in normal patients. Vertigo and dizziness have been reported rarely.
Cardiovascular :
Hypotension, palpitations, substernal pressure, phlebitis, rare cases of vasculitis, and tachycardia. (All effects due to excessively rapid infusion or insufficient dilution of drug.)
Dermatological :
Pruritus at injection site, generalised flushing, erythematous macular rash with intense pruritus over face, neck and upper body have occurred after too rapid injection of the drug. Tissue irritation and necrosis occurs after I.M. injection or extravasation from I.V. site. Vancomycin has been associated with the bullous eruption disorders, rashes including exfoliative dermatitis, Stevens-Johnson syndrome, toxic epidermal necrolysis and linear IgA bullous dermatosis. If a bullous disorder is suspected, the drug should be discontinued and specialist dermatological assessment should be carried out.
Gastrointestinal :
Oral doses are extremely unpalatable. In leukaemic patients, oral dosing regimens are associated with frequent nausea, diarrhoea and occasional vomiting.
General :
The use of Vancomycin may result in overgrowth of non-susceptible organisms resulting in new bacterial or fungal infections.
Genitourinary :
Transient elevations of urea and granular casts in the urine occasionally occur. Nephrotoxicity in the presence of normal renal function at therapeutic serum levels is rare. Rare cases of interstitial nephritis have been reported.
Haematological :
Reversible neutropenia, usually starting one week or more after onset of intravenous therapy or after a total dose of more than 25 g. Neutropenia appears to be promptly reversible when Vancomycin is discontinued. Thrombocytopenia has been rarely reported. Reversible agranulocytosis (less than 500 granulocytes per mm3) has been reported rarely, although causality has not been established. Eosinophilia has been reported.
Immunological :
Histamine release with chills, nausea, urticaria, macular rash, fever and rigors, even at normal doses but usually following rapid drug administration. Anaphylactoid reactions, hypersensitivity reactions and anaphylaxis have been reported.
Ocular :
Subconjunctival injections have infrequently been used in the treatment of bacterial corneal ulcers but may cause severe inflammation or sloughing.
Miscellaneous :
Pain and muscle spasm of the chest and back.
OVERDOSAGE :
Symptoms of overdose include ototoxicity, nephrotoxicity; there is no specific therapy for an overdosage with Vancomycin.Care is symptomatic and supportive in nature. Peritoneal filtration and haemofiltration (not dialysis) have been shown to reduce the serum concentration of Vancomycin, high flux dialysis may remove upto 25 %.
TREATMENT OF OVERDOSAGE :
Supportive care is advised with maintenance of glomerular filtration. Vancomycin is poorly removed by haemodialysis. Haemofiltration and haemoperfusion with polysuflone resin have increased Vancomycin clearance.
PHARMACEUTICAL PRECAUTIONS :
On reconstitution, parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit.
STORAGE :
Store at controlled room temperature 15 - 30°C (59 - 86°F), protected from moisture. Reconstituted solutions diluted with 5 % Dextrose injection, Sterile Water for Injection, or 0.9 % Sodium Chloride Injection is stable for 14 days if kept refrigerated at 2 - 8°C (36 - 46°F) Vancomycin solution is stable for 96 hours under refrigeration when diluted with following dilution fluids : 5 % Dextrose and normal saline, Lactated Ringers Injection, Lactated Ringers and 5 % Dextrose Injection, 5 % Dextrose, Isolyte, and Acetated Ringers Injection.
Vancomycin (10 mg / mL) diluted with Water for Injection, Sodium Chloride, or Dextrose 5 % and stored in either Becton Dickinson syringes or Braun syringes are stable (loss of potency less than 10 %) for at least 84 days in a refrigerator at 4°C (39°F).
SHELF LIFE :
24 months from the date of manufacture.
PRESENTATION :
VANCOMYCIN HYDROCHLORIDE FOR INJECTION contains Sterile Vancomycin Hydrochloride equivalent to Vancomycin 500 mg.
Single vial pack.
Disclaimer : For the use of a Registered Medical Practitioner or a Hospital or a Institution only. Also it is not intended to be used by healthcare professionals or patients for the purpose of prescribing or administering these products. Questions regarding the complete and current content of product labeling / specification / presentation should be directed to SGPharma.

 Cardiovascular
Cardiovascular