500 mg
For the use of a Registered Medical Practitioner or a Hospital or a Institution only.
Cloxacillin is an isoxazolyl penicillin and is stable to penicillinase. Chemically Cloxacillin Sodium is designated as Sodium (2S,5R,6R)-6-[[[3-(2-chlorophenyl)-5-methylisoxazol-4-yl]carbonyl]amino]-3,3-dimethyl-7-oxo-4-thia-1-azabicyclo[3.2.0]heptane-2-carboxylate.The
molecular formula C19H17CIN3NaO5S,H2O and molecular weight is 475.90.
STRUCTURAL FORMULA :
Its structural formula is :
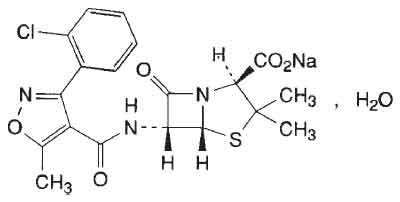
Cloxacillin Sodium for Injection B.P. 250 mg / 500 mg contains a white crystalline hygroscopic powder filled in flint tubular glass vials.
COMPOSITION :
Cloxacillin Sodium for Injection B.P. 250 mg
Each vial contains :
Sterile Cloxacillin Sodium B.P.
equivalent to Cloxacillin ................................. 250 mg
Cloxacillin Sodium for Injection B.P. 500 mg
Each vial contains :
Sterile Cloxacillin Sodium B.P.
equivalent to Cloxacillin ................................. 500 mg
ACTIONS :
Cloxacillin is bactericidal with a mode of action similar to that of benzyl penicillin. It is active against penicillinase-producing Staphylococci and in general, is less effective against organism susceptible to penicillin G e.g. Streptococci and pneumococci is not useful against
gram negative organisms.
PHARMACOKINETICS :
Cloxacillin is incompletely absorbed from the gastro-intestinal tract after oral administration and absorption is further reduced by the presence of food in the stomach. After an oral dose of 500 mg, a peak plasma concentration of 7 to 14 μg per mL is attained in fasting
subjects in 1 to 2 hours. Absorption is more complete when given by intramuscular injections and peak plasma concentrations of about 15 μg per mL have been observed 30 minutes after a dose of 500 mg. Doubling the dose can double the plasma concentration.
About 94 % of Cloxacillin in the circulation is bound to plasma proteins. Cloxacillin has been reported to have a plasma half-life of 0.5 to 1 hour. The half-life is prolonged in neonates. Cloxacillin crosses the placenta and distributed into in breast milk. There is little
diffusion into the CSF except when the meninges are inflamed. Therapeutic concentrations can be achieved in pleural and synovial fluids and in bone. Cloxacillin is metabolised to a limited extent and the unchanged drug and metabolites are excreted in the urine by glomerular filtration and renal tubular secretion. About 35 % of an oral dose is excreted in the urine and up to 10 % in the bile. Cloxacillin is not removed by haemodialysis. Plasma concentrations are enhanced if probenecid is given concomitantly. Reduced concentrations in patients with cystic fibrosis have been attributed to enhanced nonrenal clearance of Cloxacillin.
INDICATIONS :
Cloxacillin is indicated for the treatment of infections caused by penicillinase producing staphylococci that are resistant to Benzylpenicillin,
such as in :
abscesses (skin and soft tissue);
cellulitis;
bacteraemia (septicaemia);
endocarditis;
pneumonia (respiratory tract);
osteomyelitis (bone and joints).
Administration :
Please note : The reconstituted solution must be shaken well before use. Use freshly reconstituted solution.
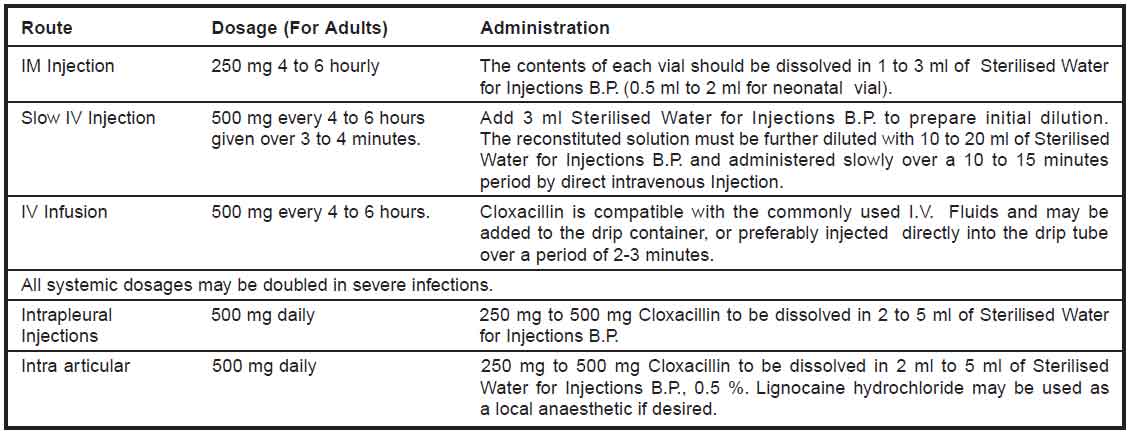
Dosage for Children : 12.5 to 25 mg / kg every 6 hours.
Under 2 years : 1/4 of adult dose.
2 to 10 years : 1/2 of adult dose.
Cloxacillin may be administered in combination with other antibacterials, including Ampicillin to produce a broader spectrum of activity.
CONTRAINDICATIONS :
Patients with a history of penicillin allergy. Neonates born of mothers sensitive to penicillin. Patients allergic to cephalosporins may also be allergic to penicillins. Cloxacillin should not be administered by subconjunctival injection or used as an eye drop. The intrathecal route should be avoided.
WARNINGS :
Use with care in jaundiced neonates. Incompatible with aminoglycosides, tetracyclines, erythromycin and polymyxin B.
Since Cloxacillin crosses the placenta, the drug should be given to a pregnant women only if clearly needed.
Nursing mothers :
Since Cloxacillin is distributed into breast milk, caution should be exercised when the drug is administered to a nursing mother.
SIDE EFFECTS :
Sensitivity reactions may include skin rashes, angioedema, bronchospasm, serum sickness and anaphylaxis and sometimes death within minutes. Treatment with adrenaline, corticosteroids, aminophyllin or antihistamine may be necessary. A generalised sensitivity reaction can develop within a few hours or weeks of commencing treatment, including urticaria, fever, joint pains and eosinophilia. Other allergic reactions include exfoliative dermatitis and other skin reactions, interstitial nephritis and vasculitis. Haemolytic anaemia, leucopenia,
prolonged bleeding time and defective platelet function. Suprainfection with C. albicans, other fungi or organisms resistant to Cloxacillin may occur. Care should be taken when administering high doses of Cloxacillin especially to patients with impaired renal function as there is a risk of neurotoxicity. Renal and haematological systems should be monitored during prolonged and high dose therapy. Patients with syphilis may exhibit the Jarish - Herxheimer reaction and should also therefore be monitored. A skin test for sensitivity may be used to determine those patients most likely to develop allergic reactions to Penicillins.
OVERDOSAGES AND TREATMENT OF OVERDOSAGE :
Disturbance of electrolyte balance may occur following administration of large doses. Convulsions and other signs of toxicity to the central nervous system may occur with very high doses, particularly when administered intravenously to patients with renal failure.
Nephrotoxicity may occur in patients with diminished renal function. Treatment of overdosage is symptomatic and supportive.
PHARMACEUTICAL PRECAUTIONS :
Solutions for injections should preferably be freshly prepared but will retain their anti-bacterial potency for 24 hours at room temperature or for 4 days at 4oC.
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration whenever solution and container permit.
STORAGE :
Store below 25oC (77oF), protected from light and moisture. Do not refrigerate.
SHELF LIFE :
24 months from the date of manufacture.
PRESENTATION :
Cloxacillin Sodium for Injection is supplied as equivalent of 250 mg & 500 mg of Cloxacillin, respectively.
10 Vials are packed in a Box.
Disclaimer : For the use of a Registered Medical Practitioner or a Hospital or a Institution only. Also it is not intended to be used by healthcare professionals or patients for the purpose of prescribing or administering these products. Questions regarding the complete and current content of product labeling / specification / presentation should be directed to SGPharma.

 Cardiovascular
Cardiovascular