4 mg/2 ml, (2 mg/2 ml as base)
For the use of a Registered Medical Practitioner or a Hospital or a Institution only.
NODRESOL (Noradrenaline) sometimes referred to as l-arterenol / levarterenol or l - norepinephrine, is a sympathomimetic amine which differs from Adrenaline by the absence of a methyl group on the nitrogen atom. Chemically, Noradrenaline Bitartrate is (+)-α-(aminomethyl)-3,4-dihydroxybenzyl alcohol tartrate (1:1) (salt), monohydrate. Its molecular formula is C8H11NO3·C4H6O6·H2O and molecular weight is 337.28.
STRUCTURAL FORMULA :
Its structural formula is :
-Structure.jpg)
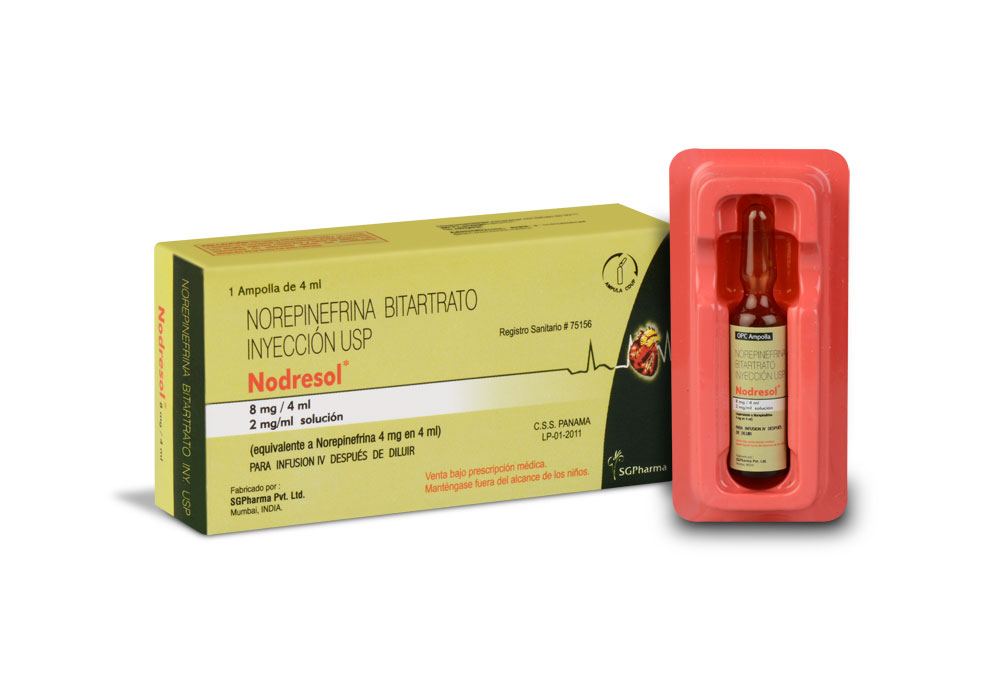
NODRESOL is a clear sterile solution filled in 2 ml amber glass ampoule.
COMPOSITION :
Each ml contains :
Noradrenaline Bitartrate I.P.
equivalent to Noradrenaline 1 mg
Water for Injections I.P. q.s.
Contains no preservatives.
ACTIONS :
Noradrenaline functions as a peripheral vasoconstrictor (alpha-adrenergic action) and as an inotropic stimulator of the heart and dilator of coronary arteries (beta-adrenergic action). These actions result in an increase in systemic blood pressure and coronary artery blood flow. In myocardial infarction accompanied by hypertension, Sterile Noradrenaline Concentrate I.P. usually increases aortic blood pressure, coronary artery blood flow and myocardial oxygenation, thereby helping to limit the area of myocardial ischaemia and infarction. Venous return is increased and the heart tends to resume a more normal rate and rhythm than in the hypotensive state. In hypotension that persists after correction of blood volume deficits, Sterile Noradrenaline Concentrate I.P. helps raise the blood pressure to an optimal level and establish a more adequate circulation.
PHARMACOKINETICS :
Absorption :
Orally ingested noradrenaline is destroyed in the GI tract and the drug is poorly absorbed after subcutaneous injection. After I.V. administration, a pressor response occurs rapidly. The drug has a short duration of action, and the pressor action stops within 1- 2 minutes after the infusion is discontinued.
Distribution :
Noradrenaline localises mainly in sympathetic nervous tissue. The drug crosses the placenta but not the blood-brain barrier.
Elimination :
The pharmacologic actions of noradrenaline are terminated primarily by uptake and metabolism in sympathetic nerve endings. The drug is metabolised in the liver and other tissues by a combination of reactions involving the enzymes catechol-O-methyltransferase (COMT) and monoamine oxidase (MAO). The major metabolites are normetanephrine and 3-methoxyl-4-hydroxy mandelic acid (vanillylmandelic acid, VMA), both of which are inactive. Other inactive metabolites include 3-methoxy-4 hydroxyphenylglycol, 3,4 hydroxymandelic acid and 3,4 dihydroxyphenylglycol. Noradrenaline metabolites are excreted in urine primarily as the sulfate conjuges and to a lesser extent, as the glucuronide conjugates. Only small quantities of noradrenaline are excreted unchanged.
INDICATIONS :
For the restoration of blood pressure in certain acute hypotensive states (e.g. phaeochromocytomectomy, sympathectomy, poliomyelitis, spinal anaesthesia, myocardial infarction, septicaemia, blood transfusion and drug reactions). As an adjunct in the treatment of cardiac arrest. To restore and maintain an adequate blood pressure after an effective heartbeat and ventilation have been established by other means.
Administration :
NODRESOL contains Noradrenaline as the acid tartrate. It is a concentrated, potent drug which must be diluted in dextrose containing solutions prior to infusion. An infusion of noradrenaline should be given into a large vein.
INSTRUCTIONS FOR USE OF AMPOULE :
The ampoule used in this product is equipped with O.P.C (One Point Cut) opening system. No ampoule file is needed to open the ampoule. The neck of the ampoule is prescored at the point of constriction. A coloured dot on the ampoule head helps to orientate the ampoule. Take the ampoule and face the coloured dot. Let the solution at the head of the ampoule to flow down by shaking or a gentle stroke. The ampoule opens easily by placing the thumb on the coloured dot and gently pressing downwards as shown.

Dosage :
Restoration of Blood Pressure in Acute Hypotensive States :
Blood volume depletion should always be corrected as fully as possible before any vasopressor is administered. When, as an emergency measure, intra-aortic pressures must be maintained to prevent cerebral or coronary artery ischaemia, noradrenaline can be administered before and concurrently with blood volume replacement.
Diluent :
NODRESOL should be diluted in 5 % Dextrose Injection I.P. or 5 % Dextrose and Sodium Chloride Injections I.P. These Dextrose containing fluids provide protection against significant loss of potency due to oxidation. Administration in saline solution alone is not recommended. Whole blood or plasma, if indicated to increase blood volume, should be administered separately (for example, by use of a Y-tube and individual containers if given simultaneously).
verage Dosage :
Add a two, 2 ml ampoules (4 ml of NODRESOL equivalent to 4 mg of noradrenaline) to 1,000 ml of a 5 % Dextrose containing solution. Each ml of this dilution contains 4 mcg of noradrenaline (or 8 mcg of the noradrenaline acid tartrate). Give this solution by intravenous infusion. Insert a plastic intravenous catheter through a suitable bore needle well advanced centrally into the vein and securely fixed with adhesive tape, avoiding, if possible, a catheter tie-intechnique as this promotes stasis. An I.V. drip chamber or other suitable metering device is essential to permit an accurate estimation of the rate of flow in drops per minute. After observing the response to an initial dose of 2 ml to 3 ml (from 8 mcg to 12 mcg of base) per minute, adjust the rate of flow to establish and maintain a low normal blood pressure (usually 80 mmHg to 100 mmHg systolic) sufficient to maintain the circulation to vital organs. In previously hypertensive patients, it is recommended that the blood pressure should be raised no higher than 40 mmHg below the pre-existing systolic pressure. The average maintenance dose ranges from 0.5 ml to 1 ml per minute (from 2 mcg to 4 mcg of base).
High Dosage :
Great individual variation occurs in the dose required to attain and maintain an adequate blood pressure. In all cases, dosage of noradrenaline should be titrated according to the response of the patient. Occasionally much larger or even enormous daily doses (as high as 68 mg base or 34 ampoules) may be necessary if the patient remains hypotensive, but occult blood volume depletion should always be suspected and corrected when present. Central venous pressure monitoring is usually helpful in detecting and treating this situation.
Fluid Intake :
The degree of dilution depends on clinical fluid volume requirements. If large volumes of fluid (dextrose) are needed at a flow rate that would involve an excessive dose of the pressor agent per unit of time, a solution more dilute than 4 mcg per ml should be used. On the other hand, when large volumes of fluid are clinically undesirable, a concentration greater than 4 mcg per ml may be necessary.
Duration of Therapy :
The infusion should be continued until adequate blood pressure and tissue perfusion are maintained without therapy. Infusions of NODRESOL should be reduced gradually, avoiding abrupt withdrawal. In some of the reported cases of vascular collapse due to acute myocardial infarction, treatment was required for up to 6 days.
Adjunctive Treatment in Cardiac Arrest :
Infusions of NODRESOL are usually administered intravenously during cardiac resuscitation to restore and maintain an adequate blood pressure after an effective heartbeat and ventilation have been established by other means. [Noradrenaline’s powerful beta-adrenergic stimulating action is also thought to increase the strength and effectiveness of systolic contractions once they occur.]
Average Dosage :
To maintain systemic blood pressure during the management of cardiac arrest, NODRESOL is used in the same manner as described under Restoration of Blood Pressure in Acute Hypotensive States.
CONTRAINDICATIONS :
NODRESOL should not be given to patients who are hypotensive from blood volume deficits except as an emergency measure to maintain coronary and cerebral artery perfusion until blood volume replacement therapy can be completed. If noradrenaline is continuously administered to maintain blood pressure in the absence of blood volume replacement, the following may occur : severe peripheral and visceral vasoconstriction, decreased renal perfusion and urine output, poor systemic blood flow despite “normal” blood pressure, tissue hypoxia, and lactate acidosis.NODRESOL should also not be given to patients with mesenteric or peripheral vascular thrombosis (because of the risk of increasing ischaemia and extending the area of infarction) unless, in the opinion of the attending physician, the administration of NODRESOL is necessary as a life-saving procedure. Cyclopropane and halothane anaesthetics increase cardiac autonomic irritability and therefore seem to sensitise the myocardium to the action of intravenously administered adrenaline Noradrenaline. Hence, the use of NODRESOL during cyclopropane and halothane anaesthesia is generally considered contraindicated because of the risk of producing ventricular tachycardia or fibrillation. The same type of cardiac arrhythmias may result from the use of NODRESOL in patients with profound hypoxia or hypercarbia.
WARNINGS :
NODRESOL should be used with extreme caution in patients receiving monoamine oxidase inhibitors (MAOI) or antidepressants of the triptyline or imipramine types, because severe, prolonged hypertension may result. NODRESOL Injection contains sodium metabisulphite, a sulphite that may cause allergic-type reactions including anaphylactic symptoms and life-threatening or less severe asthmatic episodes in certain susceptible people. The overall prevalence of sulphite sensitivity in the general population is unknown. Sulphite sensitivity is seen more frequently in asthmatic than in non-asthmatic people.
PRECAUTIONS :
General
Avoid Hypertension :
Because of the potency of noradrenaline and because of varying response to pressor substances, the possibility always exists that dangerously high blood pressure may be produced with overdoses of this pressor agent. It is desirable, therefore, to record theblood pressure every 2 minutes from the time administration is started until the desired blood pressure is obtained, then every 5 minutes if administration is to be continued. The rate of flow must be watched constantly and the patient should never be left unattended while receiving noradrenaline. Headache may be a symptom of hypertension due to overdosage.
Site of Infusion :
Whenever possible, infusions of noradrenaline should be given into a large vein, particularly an antecubital vein because, when administered into this vein, the risk of necrosis of the overlying skin from prolonged vasoconstriction is apparently very slight. Some authors have indicated that the femoral vein is also an acceptable route of administration. A catheter tie-in technique should be avoided, if possible, since the obstruction to blood flow around the tubing may cause stasis and increased local concentration of Noradrenaline. Occlusive vascular diseases (for example, atherosclerosis, arteriosclerosis, diabetic endarteritis, Buerger’s disease) are more likely to occur in the lower than in the upper extremity. Therefore, one should avoid the veins of the leg in elderly patients or in those suffering from such disorders. Gangrene has been reported in a lower extremity when infusions of Noradrenaline were given in an ankle vein.
Extravasation :
The infusion site should be checked frequently for free flow. Care should be taken to avoid extravasation of Noradrenaline into the tissues, as local necrosis might ensue due to the vasoconstrictive action of the drug. Blanching along the course of the infused vein, sometimes without obvious extravasation, has been attributed to vasa vasorum constriction with increased permeability of the vein wall, permitting some leakage. This also may progress on rare occasions to superficial slough, particularly during infusion into leg veins in elderly patients or in those suffering from obliterative vascular disease. Hence, if blanching occurs, consideration should be given to the advisability of changing the infusion site at intervals to allow the effects of local vasoconstriction to subside.
Pregnancy : Category A :
Animal reproduction studies have not been conducted with noradrenaline. It is also not known whether noradrenaline can cause foetal harm when administered to a pregnant woman or can affect reproduction capacity. Noradrenaline should be given to a pregnant woman only if clearly needed.
Nursing mothers :
It is not known whether this drug is excreted in human milk. Because many drugs are excreted in human milk, caution should be exercised when noradrenaline is administered to a nursing woman.
Paediatric Use :
Safety and effectiveness in paediatric patients has not been established.
Geriatric Use :
Clinical studies of noradrenaline dose not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. Although clinical experience has not identified differences in responses between the elderly and younger patients, dose selection for an elderly patient should be cautious, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other therapy. Noradrenaline infusions should not be administered into the veins in the leg in elderly patients.
INTERACTIONS :
Cyclopropane and halothane anaesthetics increase cardiac autonomic irritability and therefore seem to sensitise the myocardium to the action of intravenously administered adrenaline or Noradrenaline. Hence, the use of noradrenaline during cyclopropane and halothane anaesthesia is generally considered contraindicated because of the risk of producing ventricular tachycardia or fibrillation. The same type of cardiac arrhythmias may result from the use of noradrenaline in patients with profound hypoxia or hypercarbia.
Noradrenaline should be used with extreme caution in patients receiving monoamine oxidase inhibitors (MAOI) or antidepressants of the triptyline or imipramine types, because severe, prolonged hypertension may result. NODRESOL infusion solutions should not be mixed with other drugs. Infusion solutions containing noradrenaline acid tartrate have been reported to be incompatible with alkalis and oxidising agents, barbiturates, chlorpheniramine, chlorothiazide, nitrofurantoin, phenytoin, sodium bicarbonate, sodium iodide, streptomycin, sulfadiazine and sulfafurazole.
SIDE EFFECTS :
The following reactions can occur :
Body As A Whole : Ischemic injury due to potent vasoconstrictor action and tissue hypoxia.
Cardiovascular System : Bradycardia, probably as a reflex result of a rise in blood pressure, arrhythmias.
Nervous System : Anxiety, transient headache.
Respiratory System : Respiratory difficulty.
Skin and Appendages : Extravasation necrosis at injection site.
Prolonged administration of any potent vasopressor may result in plasma volume depletion which should be continuously corrected by appropriate fluid and electrolyte replacement therapy. If plasma volumes are not corrected, hypotension may recur when noradrenaline is discontinued, or blood pressure may be maintained at the risk of severe peripheral and visceral vasoconstriction (e.g. decreased renal perfusion) with diminution in blood flow and tissue perfusion with subsequent tissue hypoxia and lactic acidosis and possible ischemic injury. Gangrene of extremities has been rarely reported. Overdoses or conventional doses in hypersensitive persons (e.g. hyperthyroid patients) cause severe hypertension with violent headache, photophobia, stabbing retrosternal pain, pallor, intense sweating and vomiting.
OVERDOSAGE AND TREATMENT OF OVERDOSAGE :
Overdosage with noradrenaline may result in headache, severe hypertension, reflex bradycardia, marked increase in peripheral resistance and decreased cardiac output. In case of accidental overdosage, as evidenced by excessive blood pressure elevation, discontinue Noradrenaline until the condition of the patient stabilises.
PHARMACEUTICAL PRECAUTIONS :
Parenteral drug products should be inspected visually for particulate matter and discolouration prior to use, whenever solution and container permit. Do not use the solution if its colour is pinkish or brownish or if it contains a precipitate. Avoid contact with iron salts, alkalis, or oxidising agents.
STORAGE :
Store below 30°C (86°F), protected from light.
Do not refrigerate.
SHELF LIFE :
24 months from the date of manufacture.
PRESENTATION :
NODRESOL is supplied as 4 mg Noradrenaline Bitartrate in 2 ml aqueous solution.
Such 2 ampoules of 2 ml are packed in a Box.
Disclaimer : For the use of a Registered Medical Practitioner or a Hospital or a Institution only. Also it is not intended to be used by healthcare professionals or patients for the purpose of prescribing or administering these products. Questions regarding the complete and current content of product labeling / specification / presentation should be directed to SGPharma.

 Cardiovascular
Cardiovascular