
2 mg
For the use of a Registered Medical Practitioner or a Hospital or a Institution only.
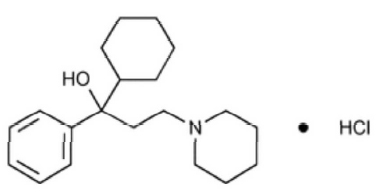
TRIHEXYPHENIDYL HYDROCHLORIDE TABLETS USP (Trihexyphenidyl Hydrochloride) is a synthetic antispasmodic drug. Chemically, Trihexyphenidyl Hydrochloride is 1-Piperidinepropanol, α-cyclohexyl-α-phenyl-, hydrochloride, (±)-. The molecular formula is C20H31NO·HCl and molecular weight is 337.93.
STRUCTURAL FORMULA :
Its structural formula is :
TRIHEXYPHENIDYL HYDROCHLORIDE TABLETS USP is white coloured, circular, biconvex tablets having breakline on one side and “SGP” embossed on other side.
COMPOSITION :
Each uncoated tablet contains :
Trihexyphenidyl Hydrochloride USP 2 mg
Excipients q.s.
ACTIONS : Trihexyphenidyl hydrochloride exerts a direct inhibitory effect upon the parasympathetic nervous system. It also has a relaxing effect on smooth musculature; exerted both directly upon the muscle tissue itself and indirectly through an inhibitory effect upon the parasympathetic nervous system. Its therapeutic properties are similar to those of atropine although undesirable side effects are ordinarily less frequent and severe than with the latter.
PHARMACOKINETICS :
TRIHEXYPHENIDYL HYDROCHLORIDE TABLETS USP is well absorbed from the gastrointestinal tract. It disappears rapidly from the plasma and tissues and does not accumulate in the body during continued administration of conventional doses.
INDICATIONS :
TRIHEXYPHENIDYL HYDROCHLORIDE TABLETS USP is indicated as an adjunct in the treatment of all forms of parkinsonism (postencephalitic, arteriosclerotic, and idiopathic). It is often useful as adjuvant therapy when treating these forms of parkinsonism with levodopa. Additionally, it is indicated for the control of extrapyramidal disorders caused by central nervous system drugs such as the dibenzoxazepines, phenothiazines, thioxanthenes, and butyrophenones.
Administration :
For oral use.
Whether trihexyphenidyl hydrochloride may best be given before or after meals should be determined by the way the patient reacts. Postencephalitic patients, who are usually more prone to excessive salivation, may prefer to take it after meals and may, in addition, require small amounts of atropine which, under such circumstances, is sometimes an effective adjuvant. If trihexyphenidyl hydrochloride tends to dry the mouth excessively, it may be better to take it before meals, unless it causes nausea. If taken after meals, the thirst sometimes induced can be allayed by mint candies, chewing gum or water.
Dosage :
Dosage should be individualized. The initial dose should be low and then increased gradually, especially in patients over 60 years of age. Abrupt withdrawal of treatment for parkinsonism may result in acute exacerbation of parkinsonism symptoms; therefore, abrupt withdrawal should be avoided. Abrupt withdrawal of treatment may result in neuroleptic malignant syndrome (NMS).
Idiopathic Parkinsonism :
As initial therapy for parkinsonism, 1 mg of trihexyphenidyl hydrochloride in tablet form may be administered the first day. The dose may then be increased by 2 mg increments at intervals of three to five days, until a total of 6 to 10 mg is given daily. The total daily dose will depend upon what is found to be the optimal level. Many patients derive maximum benefit from this daily total of 6 to 10 mg, but some patients, chiefly those in the postencephalitic group, may require a total daily dose of 12 to 15 mg.
Adults only :
Optimal dosage should always be determined empirically, usually by initiating therapy at a relatively low level and by subsequent graduated increments. The usual dosage for Parkinsonism is 6-10 mg per day although some patients chiefly in the postencephalitic group may require an average total dose of 12-15 mg daily. It should be given orally either three or four times a day at mealtimes. Normal dosage for drug-induced Parkinsonism is usually between 5 mg and 15 mg per day, although some cases have been controlled by 1 mg daily. In all cases, trihexyphenidyl hydrochloride dosage should be increased or decreased only by small increments over a period of several days. In initial therapy the dose should be 1 mg the first day, 2 mg the second day with further increases of 2 mg per day at three to five day intervals until the optimum dose is reached. If patients are already being treated with other parasympathetic inhibitors, trihexyphenidyl hydrochloride should be substituted as part of the therapy. When trihexyphenidyl hydrochloride is used concomitantly with levodopa the usual dose of each may need to be reduced. Careful adjustment is necessary, depending on side effects and the degree of symptom control. Trihexyphenidyl hydrochloride dosage of 3-6 mg daily in divided doses, is usually adequate.
Treatment of drug-induced extrapyramidal disorder :
The size and frequency of dose of trihexyphenidyl hydrochloride needed to control extrapyramidal reactions to commonly employed tranquillisers, notably the phenothiazines, thioxanthenes, and butyrophenones must be determined empirically. The total daily dosage usually ranges between 5 and 15 mg, although in some cases, these reactions have been controlled by as little as 1 mg daily. Satisfactory control may sometimes be more rapidly achieved by temporarily reducing the dosage of both drugs until the desired ataractic effect is retained without concomitant extrapyramidal reactions. It is sometimes possible to maintain the patient on reduced trihexyphenidyl hydrochloride dosage after the reactions have remained under control for several days. Since these reactions may remain in remission for long periods after discontinuation of trihexyphenidyl hydrochloride therapy, such therapy should be of minimal duration and discontinued after symptoms have subsided for a reasonable period of time.
Elderly :
Patients over 65 years of age tend to be relatively more sensitive and require smaller amounts of the drug.
Children :
Not recommended.
CONTRAINDICATIONS :
TRIHEXYPHENIDYL HYDROCHLORIDE TABLETS USP is contraindicated in patients with hypersensitivity to trihexyphenidyl hydrochloride or to any of the tablet ingredients. TRIHEXYPHENIDYL HYDROCHLORIDE TABLETS USP is also contraindicated in patients with narrow angle glaucoma. Blindness after long-term use due to narrow angle glaucoma has been reported.
Warnings :
Patients to be treated with trihexyphenidyl hydrochloride should have a gonioscope evaluation prior to initiation of therapy and close monitoring of intraocular pressures. The use of anticholinergic drugs may precipitate angle closure with an increase in intraocular pressure. If blurring of vision occurs during therapy, the possibility of narrow angle glaucoma should be considered. Blindness has been reported due to aggravation of narrow angle glaucoma. TRIHEXYPHENIDYL HYDROCHLORIDE TABLETS USP should be administered with caution in hot weather, especially when given concomitantly with other atropine-like drugs to the chronically ill, alcoholics, those who have central nervous system disease, or those who do manual labour in a hot environment. Anhidrosis may occur more readily when some disturbance of sweating already exists. If there is evidence of anhidrosis, the possibility of hyperthermia should be considered. Dosage should be decreased so that the ability to maintain body heat equilibrium via perspiration is not impaired. Severe anhidrosis and fatal hyperthermia have occurred with the use of anticholinergics under the conditions described above.
Neuroleptic Malignant Syndrome :
A potentially fatal symptom complex sometimes referred to as Neuroleptic Malignant Syndrome (NMS) has been reported in association with dose reduction or discontinuation of trihexyphenidyl hydrochloride. Clinical manifestations of NMS are hyperpyrexia, muscle rigidity, altered mental status and evidence of autonomic instability (irregular pulse or blood pressure, tachycardia, diaphoresis and cardiac dysrhythmias). The diagnostic evaluation of patients with this syndrome is complicated. In arriving at a diagnosis, it is important to identify cases where the clinical presentation includes both serious medical illness (eg, pneumonia, systemic infection, etc.) and untreated or inadequately treated extrapyramidal signs and symptoms (EPS). Other important considerations in the differential diagnosis include central anticholinergic toxicity, heat stroke, drug fever, and primary central nervous system (CNS) pathology.
Precautions :
General :
Patients with cardiac, liver, or kidney disorders, or with hypertension, should be closely monitored. Since trihexyphenidyl hydrochloride has atropine-like properties, patients on long-term treatment should be carefully monitored for untoward reactions. Since trihexyphenidyl hydrochloride has parasympatholytic activity, it should be used with caution in patients with glaucoma, obstructive disease of the gastrointestinal or genitourinary racts, and in elderly males with possible prostatic hypertrophy. Incipient glaucoma may be precipitated by parasympatholytic drugs such as trihexyphenidyl hydrochloride. Tardive dyskinesia may appear in some patients on long-term therapy with antipsychotic drugs or may occur after therapy with these drugs has been discontinued. Antiparkinsonism agents
do not alleviate the symptoms of tardive dyskinesia, and in some instances may aggravate them. However, parkinsonism and tardive dyskinesia often coexist in patients receiving chronic neuroleptic treatment, and anticholinergic therapy with trihexyphenidyl hydrochloride may relieve some of these parkinsonism symptoms. Trihexyphenidyl hydrochloride is not recommended for use in patients with tardive dyskinesia unless they have concomitant Parkinson’s disease.
Patients with arteriosclerosis or with a history of idiosyncrasy to other drugs may exhibit reactions of mental confusion, agitation, disturbed behaviour, or nausea and vomiting. Such patients should be allowed to develop a tolerance through the initial administration of a small dose and gradual increase in dose until an effective level is reached. If a severe reaction should occur, administration of the drug should be discontinued for a few days and then resumed at a lower dosage. Psychiatric disturbances can result from indiscriminate use (leading to overdosage) to sustain continued euphoria. Abrupt withdrawal of treatment for parkinsonism may result in acute exacerbation of parkinsonism symptoms; therefore, abrupt withdrawal should be avoided.
Pregnancy : Category C
Animal reproduction studies to evaluate teratogenic and embryotoxic potential have not been conducted with trihexyphenidyl hydrochloride. It is also not known whether trihexyphenidyl hydrochloride can cause foetal harm when administered to a pregnant woman or can affect reproduction capacity. Trihexyphenidyl hydrochloride should be given to a pregnant woman only if clearly needed.
Nursing mothers :
It is not known whether this drug is excreted in human milk. Because many drugs are excreted in human milk, caution should be exercised when trihexyphenidyl hydrochloride is administered to a nursing woman. As with other anticholinergics, trihexyphenidyl hydrochloride may cause suppression of lactation. Therefore, trihexyphenidyl hydrochloride should only be used if the expected benefit to the mother outweighs the potential risk to the infant.
Paediatric use :
Safety and effectiveness in paediatric patients have not been established.
INTERACTIONS AND INCOMPATIBILITIES :
Cannabinoids, barbiturates, opiates, and alcohol may have additive effects with trihexyphenidyl hydrochloride, and thus, an abuse potential exists. Concurrent use of alcohol or other CNS depressants with trihexyphenidyl hydrochloride may cause increased sedative effects. Extra care should be taken when trihexyphenidyl hydrochloride is given concomitantly with phenothiazines, clozapine, antihistamines, disopyramide, nefopam and amantadine because of the possibility of increased antimuscarinic side effects. Synergy has been reported between trihexyphenidyl hydrochloride and tricyclic antidepressants, probably because of an additive effect at the receptor site. This can cause dry mouth, constipation and blurred vision. In the elderly, there is a danger of precipitating urinary retention, acute glaucoma or paralytic ileus. Monoamine oxidase inhibitors can interact with concurrently administered anticholinergic agents including trihexyphenidyl hydrochloride. This can cause dry mouth, blurred vision, urinary hesitancy, urinary retention and constipation.
In general, anticholinergic agents should be used with caution in patients who are receiving tricyclic antidepressants or monoamine oxidase inhibitors. In patients who are already on antidepressant therapy the dose of trihexyphenidyl hydrochloride should be initially reduced and the patient reviewed regularly. Trihexyphenidyl hydrochloride may be antagonistic with the actions of metoclopramide and domperidone on gastrointestinal function. The absorption of levodopa may possibly be reduced when used in conjunction with trihexyphenidyl hydrochloride. Trihexyphenidyl hydrochloride may be antagonistic with the actions of parasympathomimetics.
SIDE EFFECTS :
Minor side effects, such as dryness of the mouth, blurred vision, dizziness, mild nausea or nervousness, will be experienced by 30 to 50 percent of all patients. These sensations, however, are much less troublesome with trihexyphenidyl hydrochloride than with belladonna alkaloids and are usually less disturbing than unalleviated parkinsonism. Such reactions tend to become less pronounced, and even to disappear, as treatment continues. Even before these reactions have remitted spontaneously, they may often be controlled by careful adjustment of dosage form, amount of drug, or interval between doses. Isolated instances of suppurative parotitis secondary to excessive dryness of the mouth, skin rashes, dilatation of the colon, paralytic ileus, and certain psychiatric manifestations such as delusions, hallucinations, and paranoia, all of which may occur with any of the atropine-like drugs, have been reported rarely with trihexyphenidyl hydrochloride.
Potential side effects associated with the use of any atropine-like drugs, including trihexyphenidyl hydrochloride, include cognitive dysfunctions, including confusion and memory impairment; constipation, drowsiness, urinary hesitancy or retention, tachycardia, dilation of the pupil, increased intraocular pressure, choreiform movements, weakness, vomiting, and headache. Exacerbation of parkinsonism with abrupt treatment withdrawal has been reported. Neuroleptic malignant syndrome with abrupt treatment withdrawal has been reported. The occurrence of angle-closure glaucoma in patients receiving trihexyphenidyl hydrochloride has been reported (blindness has been reported in some cases). Paradoxical sinus bradycardia, dry skin, and cycloplegia have been reported. In addition to adverse events seen in adults, the following adverse events have been reported in the literature in paediatric patients : hyperkinesia, psychosis, forgetfulness, weight loss, restlessness, chorea, and sleep alterations.
DRUG ABUSE AND DEPENDENCE :
Although trihexyphenidyl hydrochloride is not classified as a controlled substance, the possibility of abuse should be borne in mind due to its stimulant and euphoriant properties.
EFFECTS ON ABILITY TO DRIVE AND USE MACHINES :
Trihexyphenidyl hydrochloride can cause blurring of vision, dizziness and mild nausea. Also mental confusion in some cases.
INFORMATION FOR PATIENTS :
Trihexyphenidyl hydrochloride may impair mental and/or physical abilities required for performance of hazardous tasks, such as operating machinery or driving a motor vehicle. Patients should be cautioned about operating machinery, including automobiles, until they are reasonably certain that trihexyphenidyl hydrochloride therapy does not adversely affect their ability to engage in such activities. Because of increased sedative effects, patients should be cautioned to avoid the use of alcohol or other CNS depressants while taking trihexyphenidyl hydrochloride. Since this medication may increase the susceptibility to heat stroke (gastrointestinal (GI) problems, fever, heat intolerance), use with caution during hot weather. Patients should be advised to report the occurrence of Gl problems, fever, or heat intolerance promptly since paralytic ileus, hyperthermia, or heat stroke may occur. If GI upset occurs, trihexyphenidyl hydrochloride may be taken with food. Patients should have close monitoring of intraocular pressure.
OVERDOSAGE :
The mean oral LD50 of trihexyphenidyl hydrochloride has been reported to be 365 mg/kg (range, 325 to 410 mg/kg) in mice and 1660 mg/kg (1420 to 1940 mg/kg) in rats. At a dose of 40 mg/kg, dogs have exhibited emesis, restlessness followed by drowsiness, equilibrium disturbances, and mydriasis. In humans, doses up to 300 mg (5 mg/kg) have been ingested without fatalities or sequelae. However, rare cases of death associated with trihexyphenidyl hydrochloride overdosages taken in conjunction with other CNS-depressant agents have been reported or in patients with a compromised respiratory condition. Trihexyphenidyl hydrochloride blood concentrations associated with the fatalities ranged from 0.03 to 0.80 mg/l.
Signs and Symptoms :
Overdosage with trihexyphenidyl hydrochloride produces typical central symptoms of atropine intoxication (the central anticholinergic syndrome). Correct diagnosis depends upon recognition of the peripheral signs of parasympathetic blockade, including dilated and sluggish pupils; warm, dry skin; facial flushing; decreased secretions of the mouth, pharynx, nose, and bronchi; foul-smelling breath; elevated temperature; tachycardia, cardiac arrhythmias; decreased bowel sounds; and urinary retention. Neuropsychiatric signs such as delirium, disorientation, anxiety, hallucinations, illusions, confusion, incoherence, agitation, hyperactivity, ataxia, lip smacking and tasting movements, loss of memory, paranoia, combativeness, and seizures may be present. The condition can progress to stupor, coma, paralysis, cardiac and respiratory arrest, and death.
TREATMENT OF OVERDOSAGE :
Treatment of acute overdose involves symptomatic and supportive therapy. Gastric lavage or other methods to limit absorption should be instituted. A small dose of diazepam or a short- acting barbiturate may be administered if CNS excitation is observed. Phenothiazines are contraindicated because the toxicity may be intensified due to their antimuscarinic action, causing coma. Respiratory support, artificial respiration or vasopressor agents may be necessary. Hyperpyrexia must be reversed, fluid volume replaced and acid-balance maintained. Urinary catheterization may be necessary. It is not known if trihexyphenidyl hydrochloride is dialyzable.
STORAGE :
Stored below 30°C (86°F), protected from moisture and light. Do not refrigerate.
SHELF LIFE :
24 months from the date of manufacture.
PRESENTATION :
TRIHEXYPHENIDYL HYDROCHLORIDE TABLETS USP contains Trihexyphenidyl Hydrochloride USP 2 mg. 10 Blisters of 10 Tablets per Box.
Disclaimer : For the use of a Registered Medical Practitioner or a Hospital or a Institution only. Also it is not intended to be used by healthcare professionals or patients for the purpose of prescribing or administering these products. Questions regarding the complete and current content of product labeling / specification / presentation should be directed to SGPharma.

 Cardiovascular
Cardiovascular





