
5 mg
For the use of a Registered Medical Practitioner or a Hospital or a Institution only.
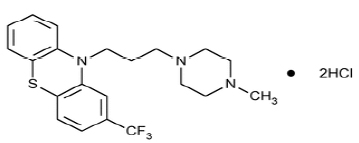
TRIFLUOPERAZINE HYDROCHLORIDE TABLETS USP (Trifluoperazine Hydrochloride) is an antipsychotic drug. Chemically, Trifluoperazine Hydrochloride is 10H -Phenothiazine, 10-[3-(4-methyl-1-piperazinyl)propyl]-2-(trifluoromethyl)-, dihydrochloride. The molecular formula is C21H24F3N3S•2HCl and molecular weight is 480.42.
STRUCTURAL FORMULA :
Its structural formula is :
TRIFLUOPERAZINE HYDROCHLORIDE TABLETS USP is green coloured, circular, biconvex film coated tablets plain on both side.
COMPOSITION :
Each film coated tablet contains :
Trifluoperazine Hydrochloride USP
equivalent to Trifluoperazine 5 mg
Excipients q.s.
Colours : Aluminium Lake Sunset Yellow, Aluminium Lake Quinoline Yellow, Aluminium Lake Brilliant Blue.
ACTIONS :
Trifluoperazine is a Piperazine Phenothiazine tranquiliser with potent anti-psychotic, anxiolytic and antiemetic activity, and a pharmacological profile of moderate sedative and hypotensive properties, and fairly pronounced tendency to cause extrapyramidal reactions.
PHARMACOKINETICS :
Trifluoperazine hydrochloride is readily absorbed from the gastrointestinal tract after oral doses; peak plasma concentrations are attained 1.5 to 6 hours after ingestion and bioavailability is subject to interindividual variation. It is highly bound to plasma proteins.
The elimination of trifluoperazine is multiphasic and the terminal half-life is about 22 hours. The major metabolite is the possibly active N-oxide. Other metabolites include the sulfoxide and the 7-hydroxy derivative. Trifluoperazine is distributed into breast milk.
INDICATIONS :
Chronic therapy :
In high doses for management of manifestations of psychotic disorders, such as acute and chronic catatonic hebephrenic and paranoid schizophrenia, psychosis due to organic brain damage, toxic psychosis, manic depressive psychosis, senile psychosis and mental deficiency.
Short-term therapy :
Treatment of acute alcoholism for the relief of delusions, hallucinations and confused state, and for the control of accompanying tremulousness and aggressive behaviour. In low doses to control excessive anxiety, tension and agitation as seen in neuroses or associated with somatic conditions. For nausea and vomiting of various causes.
Administration :
TRIFLUOPERAZINE HYDROCHLORIDE TABLETS USP is for oral administration.
Dosage :
Dosage should be tailored to the individual response, carefully monitored and adjusted accordingly. Because of the inherent long action of the drug, patients may be controlled on convenient twice daily administration.
For office patients and outpatients :
Oral – adults :
Usual dosage is 1 or 2 mg twice a day. If necessary, dosage may be increased to 6 mg a day, but above this level extrapyramidal symptoms are more likely to occur in some patients.
Oral – children :
For children of 3 - 5 years of age, up to 1 mg a day in divided doses. For children 6 - 12 years of age, the dosage may be increased to a maximum of 4 mg a day according to body weight and general physical condition. Dosage is based on a rate of 1 mg per 20 kg body weight per day.
For hospitalised patients or those under close supervision :
Oral – adults :
Usual starting dosage is 2 mg to 5 mg twice daily. The recommended starting dosage for physically fit adults is 5 mg twice a day. Small or emaciated patients should always be started on a lower dosage. After a week, this may be increased to 15 mg a day in divided doses. If necessary, further increases of 5 mg may be made at 3-day intervals, but not more often. Most patients will show optimum response on 15 to 20 mg daily, although a few will require more. When satisfactory control has been achieved, dosage may be reduced gradually until an effective maintenance level has been established.
Oral – children (6 – 12 years) :
The starting dosage is 1 mg twice daily. Any subsequent increase should be made with caution at intervals of not less than 3 days and taking into account age, body weight and severity of symptoms. It is usually not necessary to exceed dosages of 15 mg daily.
CONTRAINDICATIONS :
TRIFLUOPERAZINE HYDROCHLORIDE TABLETS USP is contraindicated in :
- cases of known hypersensitivity to trifluoperazine or excipients.
- comatose states.
- the presence of large amounts of central nervous system depressants (alcohol, barbiturates, opiates, etc.).
- circulatory collapse.
- phaeochromocytoma.
- blood dyscrasias, liver disease or bone marrow depression.
TRIFLUOPERAZINE HYDROCHLORIDE TABLETS USP contains lactose which is contra-indicated in patients with galactosaemia, the glucose-galactose malabsorption syndrome, or lactase deficiency.
WARNINGS :
Increased Mortality in Elderly Patients with Dementia-Related Psychosis :
Elderly patients with dementia-related psychosis treated with antipsychotic drugs are at an increased risk of death. Trifluoperazine hydrochloride is not approved for the treatment of patients with dementia related psychosis (see BOXED WARNING).
Tardive Dyskinesia :
Tardive dyskinesia, a syndrome consisting of potentially irreversible, involuntary, dyskinetic movements, may develop in patients treated with antipsychotic drugs. Although the prevalence of the syndrome appears to be highest among the elderly, especially elderly women, it is impossible to rely upon prevalence estimates to predict, at the inception of antipsychotic treatment, which patients are likely to develop the syndrome. Whether antipsychotic drug products differ in their potential to cause tardive dyskinesia is unknown. Both the risk of developing the syndrome and the likelihood that it will become irreversible are believed to increase as the duration of treatment and the total cumulative dose of antipsychotic drugs administered to the patient increase. However, the syndrome can develop, although much less commonly, after relatively brief treatment periods at low doses. There is no known treatment for established cases of tardive dyskinesia, although the syndrome may remit, partially or completely, if antipsychotic treatment is withdrawn. Antipsychotic treatment itself, however, may suppress (or partially suppress) the signs and symptoms of the syndrome and thereby may possibly mask the underlying disease process. The effect that symptomatic suppression has upon the long-term course of the syndrome is unknown.
Given these considerations, antipsychotics should be prescribed in a manner that is most likely to minimize the occurrence of tardive dyskinesia. Chronic antipsychotic treatment should generally be reserved for patients who suffer from a chronic illness that 1) is known to respond to antipsychotic drugs, and, 2) for whom alternative, equally effective, but potentially less harmful treatments are not available or appropriate. In patients who do require chronic treatment, the smallest dose and the shortest duration of treatment producing a satisfactory clinical response should be sought. The need for continued treatment should be reassessed periodically. If signs and symptoms of tardive dyskinesia appear in a patient on antipsychotics, drug discontinuation should be considered. However, some patients may require treatment despite the presence of the syndrome.
Neuroleptic Malignant Syndrome (NMS) :
A potentially fatal symptom complex sometimes referred to as Neuroleptic Malignant Syndrome (NMS) has been reported in association with antipsychotic drugs. Clinical manifestations of NMS are hyperpyrexia, muscle rigidity, altered mental status and evidence of autonomic instability (irregular pulse or blood pressure, tachycardia, diaphoresis, and cardiac dysrhythmias). The diagnostic evaluation of patients with this syndrome is complicated. In arriving at a diagnosis, it is important to identify cases where the clinical presentation includes both serious medical illness (e.g., pneumonia, systemic infection, etc.) and untreated or in adequately treated extra pyramidal signs and symptoms (EPS). Other important considerations in the differential diagnosis include central anticholinergic toxicity, heat stroke, drug fever and primary central nervous system (CNS) pathology. The management of NMS should include 1) immediate discontinuation of antipsychotic drugs and other drugs not essential to concurrent therapy, 2) intensive symptomatic treatment and medical monitoring, and 3) treatment of any concomitant serious medical problems for which specific treatments are available. There is no general agreement about specific pharmacological treatment regimens for uncomplicated NMS. If a patient requires antipsychotic drug treatment after recovery from NMS, the potential reintroduction of drug therapy should be carefully considered. The patient should be carefully monitored, since recurrences of NMS have been reported.
An encephalopathic syndrome (characterized by weakness, lethargy, fever, tremulousness and confusion, extrapyramidal symptoms, leukocytosis, elevated serum enzymes, BUN and FBS) has occurred in a few patients treated with lithium plus an antipsychotic. In some instances, the syndrome was followed by irreversible brain damage. Because of a possible causal relationship between these events and the concomitant administration of lithium and antipsychotics, patients receiving such combined therapy should be monitored closely for early evidence of neurologic toxicity and treatment discontinued promptly if such signs appear. This encephalopathic syndrome may be similar to or the same as neuroleptic malignant syndrome (NMS). Patients who have demonstrated a hypersensitivity reaction (e.g., blood dyscrasias, jaundice) with a phenothiazine should not be reexposed to any phenothiazine, including trifluoperazine hydrochloride, unless in the judgment of the physician the potential benefits of treatment outweigh the possible hazard. Trifluoperazine hydrochloride may impair mental and/or physical abilities, especially during the first few days of therapy. Therefore, caution patients about activities requiring alertness (e.g., operating vehicles or machinery). If agents such as sedatives, narcotics, anaesthesia, tranquilizers or alcohol are used either simultaneously or successively with the drug, the possibility of an undesirable additive depressant effect should be considered.
PRECAUTIONS :
General :
Given the likelihood that some patients exposed chronically to antipsychotics will develop tardive dyskinesia, it is advised that all patients in whom chronic use is contemplated be given, if possible, full information about this risk. The decision to inform patients and/or their guardians must obviously take into account the clinical circumstances and the competency of the patient to understand the information provided. Thrombocytopenia and anaemia have been reported in patients receiving the drug. Agranulocytosis and pancytopenia have also been reported–warn patients to report the sudden appearance of sore throat or other signs of infection. If white blood cell and differential counts indicate cellular depression, stop treatment and start antibiotic and other suitable therapy. Jaundice of the cholestatic type of hepatitis or liver damage has been reported. If fever with grippe-like symptoms occurs, appropriate liver studies should be conducted. If tests indicate an abnormality, stop treatment. One result of therapy may be an increase in mental and physical activity. For example, a few patients with angina pectoris have complained of increased pain while taking the drug. Therefore, angina patients should be observed carefully and, if an unfavorable response is noted, the drug should be withdrawn.
Because hypotension has occurred, large doses and parenteral administration should be avoided in patients with impaired cardiovascular systems. To minimize the occurrence of hypotension after injection, keep patient lying down and observe for at least 1/2 hour. If hypotension occurs from parenteral or oral dosing, place patient in head-low position with legs raised. If a vasoconstrictor is required, norepinephrine bitartrate and phenylephrine hydrochloride are suitable. Other pressor agents, including epinephrine, should not be used as they may cause a paradoxical further lowering of blood pressure. Since certain phenothiazines have been reported to produce retinopathy, the drug should be discontinued if ophthalmoscopic examination or visual field studies should demonstrate retinal changes. An antiemetic action of trifluoperazine hydrochloride may mask the signs and symptoms of toxicity or overdosage of other drugs and may obscure the diagnosis and treatment of other conditions such as intestinal obstruction, brain tumour and Reye’s syndrome.
With prolonged administration at high dosages, the possibility of cumulative effects, with sudden onset of severe central nervous system or vasomotor symptoms, should be kept in mind.
Antipsychotic drugs elevate prolactin levels; the elevation persists during chronic administration. Tissue culture experiments indicate that approximately 1/3 of human breast cancers are prolactin-dependent in vitro, a factor of potential importance if the prescribing of these drugs is contemplated in a patient with a previously detected breast cancer. Although disturbances such as galactorrhoea, amenorrhoea, gynaecomastia and impotence have been reported, the clinical significance of elevated serum prolactin levels is unknown for most patients. An increase in mammary neoplasms has been found in rodents after chronic administration of antipsychotic drugs. Neither clinical nor epidemiologic studies conducted to date, however, have shown an association between chronic administration of these drugs and mammary tumorigenesis; the available evidence is considered too limited to be conclusive at this time. Chromosomal aberrations in spermatocytes and abnormal sperm have been demonstrated in rodents treated with certain antipsychotics. Because phenothiazines may interfere with thermoregulatory mechanisms, use with caution in persons who will be exposed to extreme heat. As with all drugs which exert an anticholinergic effect, and/or cause mydriasis, trifluoperazine should be used with caution in patients with glaucoma.
Phenothiazines may diminish the effect of oral anticoagulants. Phenothiazines can produce alpha-adrenergic blockade. Concomitant administration of propranolol with phenothiazines results in increased plasma levels of both drugs. Antihypertensive effects of guanethidine and related compounds may be counteracted when phenothiazines are used concurrently. Thiazide diuretics may accentuate the orthostatic hypotension that may occur with phenothiazines.
Phenothiazines may lower the convulsive threshold; dosage adjustments of anticonvulsants may be necessary. Potentiation of anticonvulsant effects does not occur. However, it has been reported that phenothiazines may interfere with the metabolism of phenytoin and thus precipitate phenytoin toxicity. Drugs which lower the seizure threshold, including phenothiazine derivatives, should not be used with metrizamide. As with other phenothiazine derivatives, trifluoperazine hydrochloride should be discontinued at least 48 hours before myelography, should not be resumed for at least 24 hours post procedure and should not be used for the control of nausea and vomiting occurring either prior to myelography or post procedure with metrizamide.
Pregnancy :
Safety for the use of trifluoperazine hydrochloride during pregnancy has not been established. Therefore, it is not recommended that the drug be given to pregnant patients except when, in the judgment of the physician, it is essential. The potential benefits should clearly outweigh possible hazards. There are reported instances of prolonged jaundice, extrapyramidal signs, hyperreflexia or hyporeflexia in newborn infants whose mothers received phenothiazines. Reproductive studies in rats given over 600 times the human dose showed an increased incidence of malformations above controls and reduced litter size and weight linked to maternal toxicity. These effects were not observed at half this dosage. No adverse effect on Foetal development was observed in rabbits given 700 times the human dose nor in monkeys given 25 times the human dose.
Nonteratogenic Effects :
Neonates exposed to antipsychotic drugs, during the third trimester of pregnancy are at risk for extrapyramidal and/or withdrawal symptoms following delivery. There have been reports of agitation, hypertonia, hypotonia, tremor, somnolence, respiratory distress and feeding disorder in these neonates. These complications have varied in severity; while in some cases symptoms have been self-limited, in other cases neonates have required intensive care unit support and prolonged hospitalization. Trifluoperazine hydrochloride should be used during pregnancy only if the potential benefit justifies the potential risk to the Foetus.
Nursing mothers :
There is evidence that phenothiazines are excreted in the breast milk of nursing mothers. Because of the potential for serious adverse reactions in nursing infants from trifluoperazine, a decision should be made whether to discontinue nursing or to discontinue the drug, taking into account the importance of the drug to the mother.
Paediatric Use :
Safety and effectiveness not established in children younger than 6 years.
INTERACTIONS AND INCOMPATIBILITIES :
Trifluoperazine can increase the central nervous system depression produced by other CNS-depressant drugs including alcohol, hypnotics, sedatives or strong analgesics. Trifluoperazine may antagonise the action of adrenaline and other sympathomimetic agents and reverses the blood-pressure-lowering effects of adrenergic-blocking agents such as guanethidine and clonidine. Trifluoperazine may impair the metabolism of tricyclic antidepressants, the anti-Parkinson effects of levodopa and the effects of anticonvulsants.
Trifluoperazine may possibly affect the control of diabetes, or the action of anticoagulants. Antacids can impair absorption. Tea and coffee may prevent absorption by causing insoluble precipitates. Undesirable anticholinergic effects can be enhanced by anti-Parkinsonian medications (e.g. benzhexol) or other anticholinergic drugs (e.g. orphenedrine, benztropine). Phenothiazines may enhance the cardiac-depressant effects of quinidine, the absorption of corticosteroids and digoxin, the effect of diazoxide and of neuromuscular blocking agents. Trifluoperazine may interact with anti-diabetic drugs. The possibility of interaction with lithium should be borne in mind. Desferrioxamine should not be used in combination with Trifluoperazine.
Laboratory Test Interactions :
False-positive pregnancy tests may occur but are less likely to occur with serum test. False-positive test for phenylketonuria may occur.
SIDE EFFECTS :
Drowsiness, sedation, dry mouth and nasal stuffiness may occur, particularly with high dosage and at the start of treatment. Dose-related postural hypotension may occur, particularly in the elderly and after intramuscular injections. Other dose-related anticholinergic-type side effects include blurring of vision, tachycardia, constipation and urinary hesitancy or retention. Trifluoperazine may impair alertness, especially at the start of treatment. These effects may be potentiated by alcohol. Extrapyramidal reactions are common and sometimes occur at low dosage. Acute dystonias may occur early in treatment. Parkinsonian rigidity, tremor, akathisia tend to appear less rapidly. Oculogyric crises have been reported. Anti-Parkinson agents should not be prescribed routinely, because of the possible risks of aggravating anticholinergic side effects of Trifluoperazine, of precipitating toxic confusional states or of impairing its therapeutic efficacy. They should only be given as required. Tardive dyskinesia is a syndrome of irregularly repetitive involuntary movement, which may occur during administration or after withdrawal of Trifluoperazine and other neuroleptic drugs. It is characterised by abnormal writhing movements or protrusions of the tongue with lip-smacking, packering and chewing movements and facial grimaces. Choreoathetoid movements of the extremities or repetitive movements of the neck or trunk may accompany the orofacial dyskinesia or can occur alone. The syndrome is common among patients treated with moderate to high doses of antipsychotic drugs for prolonged periods of time and may prove irreversible, particularly in patients over the age of 50.
It is unlikely to occur in the short term when low or moderate doses are used as recommended, but tardive dyskinesia has been reported even when low doses of Trifluoperazine have been used for a few months. Since its occurrence may be related to duration of treatment as well as daily dose, Trifluoperazine should be given in the minimal effective dose for the minimum possible time, unless it is established that long-term administration for the treatment of schizophrenia is required. The potential seriousness and unpredictability of tardive dyskinesia and the fact that it has occasionally been reported to occur when neuroleptic antipsychotic drugs have been prescribed for relatively short periods in low dosage means that the prescribing of such agents requires especially careful assessment of risks versus benefit. Tardive dyskinesia can be precipitated or aggravated by anti-Parkinson drugs. Short-lived dyskinesias may occur after abrupt drug withdrawal. In schizophrenia, the response to antipsychotic drug treatment may be delayed. If drugs are withdrawn, recurrence of symptoms may not become apparent for several weeks or months.
Trifluoperazine, even in low dosage in susceptible (especially non-psychotic) individuals, may cause unpleasant subjective feelings of being mentally dulled or slowed down, nausea, dizziness, headache or paradoxical effects of excitement, agitation-or insomnia.
Confusional states or epileptic fits can occur. The elderly are more susceptible to the sedative and hypotensive effects. The effects of phenothiazines on the heart are dose-related ECG changes, with prolongation of the QT interval and T-wave changes have been reported commonly in patients treated with moderate to high dosage; they are reversible on reducing the dose. In a very small number of cases, they have been reported to precede serious arrhythmias, including ventricular tachycardia and fibrillation, which have also occurred after overdosage. Hormonal effects of antipsychotic neuroleptic drugs include hyperprolactinaemia, which may cause galactorrhoea, gynaecomastia and oligo-or amenorrhoea. Sexual function, including erection and ejaculation is sometimes impaired by Trifluoperazine. Weight gain may occur. Oedema has been reported with phenothiazine medication. These effects may be prevented by reduction in dosage. Raised serum cholesterol and, rarely, hyperglycaemia have been reported in association with phenothiazines.
Blood Dyscrasias : Agranulocytosis has been reported very rarely, most commonly in the first three months of treatment, but occasionally later. Blood counts should be performed if the patient develops signs of a persistent infection. Transient leucopenia can also occur.
Trifluoperazine, rarely, causes increased susceptibility to sunburn and patients should be warned to avoid excessive exposure. Skin rashes have occurred rarely. The occurrence of lenticular opacities has been reported. Trifluoperazine may impair body temperature-regulation and cases of severe hypothermia or hyperpyrexia have been reported, usually in association with moderate or high dosage of phenothiazines. The elderly or hypothyroid patient may be particularly susceptible to hyperthermia. The hazard of hyperthermia may be increased by especially hot or humid weather or by drugs, such as anti-Parkinson agents, which impair sweating. Trifluoperazine can, very rarely, cause obstructive jaundice associated with stasis in billiary canaliculi. It has been thought to be a hypersensitivity reaction. Transient abnormalities of liver function tests may occur in the absence of jaundice. Neuroleptic malignant syndrome is a rare but occasionally fatal complication of treatment with various neuroleptic drugs and is characterized by hyperpyrexia, muscle rigidity, altered consciousness and autonomic instability. Intensive symptomatic treatment, following discontinuation of trifluoperazine, should include cooling. Intravenous dantrolene has been suggested for muscle rigidity. With long-term usage, very rarely Trifluoperazine can cause increased melanin pigmentation of the skin, which eventually may develop a bluish-grey colouration. Pigment deposits also occur in the eye and other tissues. Permanent deposits, leading to impairment of vision, may develop in the lens. Epithelial keratophathy has been reported. Acute withdrawal symptoms including nausea, vomiting and insomnia have very rarely been described alter abrupt cessation of high doses of phenothazines. Gradual withdrawal is advisable.
EFFECT ON ABILITY TO DRIVE AND USE MACHINES :
TRIFLUOPERAZINE HYDROCHLORIDE TABLETS USP may cause side effects including drowsiness, dizziness and visual disturbances which interfere with the ability to drive and operate machinery. Do not drive or use machines when you first start to take this medicine until you are certain that you are not getting these side effects.
INFORMATION FOR PATIENTS :
- Advise all patients in whom long-term use is contemplated about the risk of developing tardive dyskinesia and/or NMS.
- Instruct patient to avoid alcoholic beverages and other depressants while taking this medication.
- Advise patient that this drug may cause drowsiness or impaired judgment or thinking skills and to use caution while driving or performing other tasks requiring mental alertness until tolerance is determined.
- Advise patients to report sudden appearance of sore throat or other signs of infection.
OVERDOSAGE :
Symptoms of overdose are primarily extrapyramidal reactions and symptoms of central nervous system depression. Agitation and restlessness may also occur. Other possible manifestations include convulsions, ECG changes and cardiac arrhythmias, fever, and autonomic reactions such as hypotension, dry mouth, and ileus.
TREATMENT OF OVERDOSAGE :
Treatment is essentially symptomatic and supportive. Early gastric lavage is recommended. Keep patient under observation and maintain an open airway, since involvement of the extrapyramidal mechanism may produce dysphagia and respiratory difficulty in severe overdosage. Do not attempt to induce emesis because a dystonic reaction of the head or neck may develop that could result in aspiration of vomitus.
For treatment of hypotension :
Extrapyramidal symptoms may be treated with anti-Parkinson drugs, barbiturates or diphenhydramine. If administration of a stimulant is desirable, amphetamine, dextroamphetamine or caffeine with sodium benzoate is recommended. Stimulants that may cause convulsions (e.g. picrotoxin or pentylenetetrazol) should be avoided. If hypotension occurs, the standard measures for managing circulatory shock should be initiated. If a vasoconstrictor is required, noradrenaline and phenylephrine are most suitable. Other pressor agents, including adrenaline, are not recommended because phenothiazine derivatives may reverse the usual elevating action of these agents and cause a further lowering of blood pressure. Limited experience indicates that phenothiazines are not dialyzable.
STORAGE :
Store below 30°C (86°F), protected from moisture and light. Do not refrigerate.
SHELF LIFE :
24 months from the date of manufacture.
PRESENTATION :
TRIFLUOPERAZINE HYDROCHLORIDE TABLETS USP contains Trifluoperazine Hydrochloride USP equivalent to Trifluoperazine 5 mg. 3 Blisters of 10 Tablets per Box.
Disclaimer : For the use of a Registered Medical Practitioner or a Hospital or a Institution only. Also it is not intended to be used by healthcare professionals or patients for the purpose of prescribing or administering these products. Questions regarding the complete and current content of product labeling / specification / presentation should be directed to SGPharma.

 Cardiovascular
Cardiovascular





