
75 mg, 100 mg,
150 mg, 300 mg
For the use of a Registered Medical Practitioner or a Hospital or a Institution only.
PREGABALIN CAPSULES I.P. (Pregabalin) is an antiepileptic drug. Chemically, Pregabalin is (S)-4-amino-3-(2-methylpropyl)butyric acid. The molecular formula is C8H17NO2 and molecular weight is 159.2.
STRUCTURAL FORMULA :
Its structural formula is :
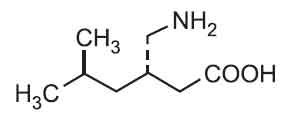
PREGABALIN CAPSULES I.P. are hard gelatin capsules containing white crystalline powder.
COMPOSITION :
Each hard gelatin capsule contains :
Pregabalin I.P. 75 mg
Excipients q.s.
Approved colours used in empty capsule shells.
Each hard gelatin capsule contains :
Pregabalin I.P. 100 mg
Excipients q.s.
Approved colours used in empty capsule shells.
Each hard gelatin capsule contains :
Pregabalin I.P. 150 mg
Excipients q.s.
Approved colours used in empty capsule shells.
Each hard gelatin capsule contains :
Pregabalin I.P. 300 mg
Excipients q.s.
Approved colours used in empty capsule shells.
ACTIONS :
Pregabalin binds with high affinity to the alpha2-delta site (an auxiliary subunit of voltage-gated calcium channels) in central nervous system tissues. Although the mechanism of action of pregabalin has not been fully elucidated, results with genetically modified mice and with compounds structurally related to pregabalin (such as gabapentin) suggest that binding to the alpha2-delta subunit may be involved in pregabalin’s anti-nociceptive and antiseizure effects in animals. In animal models of nerve damage, pregabalin has been shown to reduce calcium-dependent release of pro-nociceptive neurotransmitters in the spinal cord, possibly by disrupting alpha2-delta containing-calcium channel trafficking and/or reducing calcium currents. Evidence from other animal models of nerve damage and persistent pain suggest the anti-nociceptive activities of pregabalin may also be mediated through interactions with descending noradrenergic and serotonergic pathways originating from the brainstem that modulate pain transmission in the spinal cord.
While pregabalin is a structural derivative of the inhibitory neurotransmitter gamma-aminobutyric acid (GABA), it does not bind directly to GABAA, GABAB, or benzodiazepine receptors, does not augment GABAA responses in cultured neurons, does not alter rat brain GABA concentration or have acute effects on GABA uptake or degradation. However, in cultured neurons prolonged application of pregabalin increases the density of GABA transporter protein and increases the rate of functional GABA transport. Pregabalin does not block sodium channels, is not active at opiate receptors, and does not alter cyclooxygenase enzyme activity. It is inactive at serotonin and dopamine receptors and does not inhibit dopamine, serotonin, or noradrenaline reuptake.
PHARMACOKINETICS :
Pregabalin is rapidly absorbed after oral doses and peak plasma concentrations are achieved within 1.5 hours. Oral bioavailability is about 90 %. The rate but not the extent of absorption is reduced if given with food but this is not clinically significant. Steady state is achieved after 1 to 2 days. Pregabalin is not bound to plasma proteins and undergoes negligible metabolism. About 98 % of a dose is excreted in the urine as unchanged drug. The mean elimination half-life is 6.3 hours. Pregabalin is removed by haemodialysis.
INDICATIONS :
Neuropathic pain :
PREGABALIN CAPSULES I.P. is indicated for the treatment of peripheral and central neuropathic pain in adults.
Epilepsy :
PREGABALIN CAPSULES I.P. is indicated as adjunctive therapy in adults with partial seizures with or without secondary generalisation.
Generalised Anxiety Disorder :
PREGABALIN CAPSULES I.P. is indicated for the treatment of Generalised Anxiety Disorder (GAD) in adults.
Administration :
PREGABALIN CAPSULES I.P. is for oral use only. PREGABALIN CAPSULES I.P. may be taken with or without food.
Dosage :
The dose range is 150 to 600 mg per day given in either two or three divided doses.
Neuropathic pain :
PREGABALIN CAPSULES I.P. treatment can be started at a dose of 150 mg per day given as two or three divided doses. Based on individual patient response and tolerability, the dose may be increased to 300 mg per day after an interval of 3 to 7 days, and if needed, to a maximum dose of 600 mg per day after an additional 7-day interval.
Epilepsy :
PREGABALIN CAPSULES I.P. treatment can be started with a dose of 150 mg per day given as two or three divided doses. Based on individual patient response and tolerability, the dose may be increased to 300 mg per day after 1 week. The maximum dose of 600 mg per day may be achieved after an additional week.
Generalised Anxiety Disorder :
The dose range is 150 to 600 mg per day given as two or three divided doses. The need for treatment should be reassessed regularly. PREGABALIN CAPSULES I.P. treatment can be started with a dose of 150 mg per day. Based on individual patient response and tolerability, the dose may be increased to 300 mg per day after 1 week. Following an additional week the dose may be increased to 450 mg per day. The maximum dose of 600 mg per day may be achieved after an additional week.
Discontinuation of pregabalin :
In accordance with current clinical practice, if pregabalin has to be discontinued it is recommended this should be done gradually over a minimum of 1 week independent of the indication.
Patients with renal impairment :
Pregabalin is eliminated from the systemic circulation primarily by renal excretion as unchanged drug. As pregabalin clearance is directly proportional to creatinine clearance, dose reduction in patients with compromised renal function must be individualised according to creatinine clearance (CLcr).
Patients with hepatic impairment :
No dose adjustment is required for patients with hepatic impairment.
Paediatric population :
The safety and efficacy of PREGABALIN CAPSULES I.P. in children below the age of 12 years and in adolescents (12 - 17 years of age) have not been established. No data are available.
Elderly (over 65 years of age) population :
Elderly patients may require a dose reduction of pregabalin due to a decreased renal function.
CONTRAINDICATIONS :
PREGABALIN CAPSULES I.P. is contraindicated in patients with known hypersensitivity to pregabalin or any of its components. Angioedema and hypersensitivity reactions have occurred in patients receiving pregabalin therapy. PREGABALIN CAPSULES I.P. is contraindicated during breastfeeding. PREGABALIN CAPSULES I.P. contains lactose which is contra-indicated in patients with galactosaemia, the glucose-galactose malabsorption syndrome, or lactase deficiency.
Diabetic patients :
PREGABALIN CAPSULES I.P. should be used cautiously in diabetic patients. In accordance with current clinical practice, some diabetic patients who gain weight on PREGABALIN CAPSULES I.P. treatment may need to adjust hypoglycaemic medicinal products.
Hypersensitivity reactions :
There have been reports in the postmarketing experience of hypersensitivity reactions, including cases of angioedema. PREGABALIN CAPSULES I.P. should be discontinued immediately if symptoms of angioedema, such as facial, perioral, or upper airway swelling occur.
Dizziness, somnolence, loss of consciousness, confusion, and mental impairment :PREGABALIN CAPSULES I.P. treatment has been associated with dizziness and somnolence, which could increase the occurrence of accidental injury (fall) in the elderly population. There have also been post-marketing reports of loss of consciousness, confusion and mental impairment. Therefore, patients should be advised to exercise caution until they are familiar with the potential effects of the medicinal product.
Vision-related effects :
In controlled trials, a higher proportion of patients treated with PREGABALIN CAPSULES I.P. reported blurred vision than did patients treated with placebo which resolved in a majority of cases with continued dosing. In the clinical studies where ophthalmologic testing was conducted, the incidence of visual acuity reduction and visual field changes was greater in PREGABALIN CAPSULES I.P. treated patients than in placebo-treated patients; the incidence of fundoscopic changes was greater in placebo-treated patients. In the post-marketing experience, visual adverse reactions have also been reported, including loss of vision, visual blurring or other changes of visual acuity, many of which were transient. Discontinuation of PREGABALIN CAPSULES I.P. may result in resolution or improvement of these visual symptoms.
Renal failure :
Cases of renal failure have been reported and in some cases discontinuation of PREGABALIN CAPSULES I.P. did show reversibility of this adverse reaction.
Withdrawal of concomitant antiepileptic medicinal products :
There are insufficient data for the withdrawal of concomitant antiepileptic medicinal products, once seizure control with PREGABALIN CAPSULES I.P. in the add-on situation has been reached, in order to reach monotherapy on PREGABALIN CAPSULES I.P.
Withdrawal symptoms :
After discontinuation of short-term and long-term treatment with PREGABALIN CAPSULES I.P. withdrawal symptoms have been observed in some patients. The following events have been mentioned : insomnia, headache, nausea, anxiety, diarrhoea, flu syndrome, nervousness, depression, pain, convulsion, hyperhidrosis and dizziness. The patient should be informed about this at the start of the treatment. Convulsions, including status epilepticus and grand mal convulsions, may occur during PREGABALIN CAPSULES I.P. use or shortly after discontinuing PREGABALIN CAPSULES I.P. Concerning discontinuation of long-term treatment of PREGABALIN CAPSULES I.P., data suggest that the incidence and severity of withdrawal symptoms may be dose-related.
Congestive heart failure :
There have been post-marketing reports of congestive heart failure in some patients receiving pregabalin. These reactions are mostly seen in elderly cardiovascular compromised patients during PREGABALIN CAPSULES I.P. treatment for a neuropathic indication. PREGABALIN CAPSULES I.P. should be used with caution in these patients. Discontinuation of PREGABALIN CAPSULES I.P. may resolve the reaction. Treatment of central neuropathic pain due to spinal
cord injury :
In the treatment of central neuropathic pain due to spinal cord injury the incidence of adverse reactions in general, central nervous system adverse reactions and especially somnolence was increased. This may be attributed to an additive effect due to concomitant medicinal products (e.g. anti-spasticity agents) needed for this condition. This should be considered when prescribing PREGABALIN CAPSULES I.P. in this condition.
Suicidal ideation and behaviour :
Suicidal ideation and behaviour have been reported in patients treated with anti-epileptic agents in several indications. A meta-analysis of randomised placebo controlled studies of anti-epileptic drugs has also shown a small increased risk of suicidal ideation and behaviour. The mechanism of this risk is not known and the available data do not exclude the possibility of an increased risk for pregabalin. Therefore patients should be monitored for signs of suicidal ideation and behaviours and appropriate treatment should be considered. Patients (and caregivers of patients) should be advised to seek medical advice should signs of suicidal ideation or behaviour emerge.
Reduced lower gastrointestinal tract function :
There are post-marketing reports of events related to reduced lower gastrointestinal tract function (e.g., intestinal obstruction, paralytic ileus, constipation) when PREGABALIN CAPSULES I.P. was co-administered with medications that have the potential to produce constipation, such as opioid analgesics. When PREGABALIN CAPSULES I.P. and opioids will be used in combination, measures to prevent constipation may be considered (especially in female patients and elderly).
Abuse potential :
Cases of abuse have been reported. Caution should be exercised in patients with a history of substance abuse and the patient should be monitored for symptoms of pregabalin abuse.
Encephalopathy :
Cases of encephalopathy have been reported, mostly in patients with underlying conditions that may precipitate encephalopathy.
Pregnancy : Category B3
Pregabalin has not been studied in pregnant women and pregabalin should not be used during pregnancy unless the benefit to the mother clearly outweighs the potential risk to the foetus. In a pre- and post-natal study in rats, pregabalin treatment resulted in offspring developmental toxicity at exposures (plasma AUC) > 5 times the expected human exposure at the maximum recommended clinical dose of 600 mg/day. Offspring development was unaffected at 2 times the expected maximum human exposure.
Nursing Mothers :
It is not known if pregabalin is excreted in the breast milk of humans; however, it is present in the milk of rats. Therefore, breastfeeding is not recommended during treatment with pregabalin.
Paediatric Use :
Safety and efficacy of pregabalin in paediatric patients have not been established.
EFFECTS ON ABILITY TO DRIVE AND USE MACHINES :
PREGABALIN CAPSULES I.P. may cause dizziness and somnolence and therefore may influence the ability to drive or use machines. Patients are advised not to drive, operate complex machinery or engage in other potentially hazardous activities until it is known whether this medicinal product affects their ability to perform these activities.
INTERACTIONS AND INCOMPATIBILITIES :
Since pregabalin is predominantly excreted unchanged in the urine, undergoes negligible metabolism in humans (< 2 % of a dose recovered in urine as metabolites), does not inhibit drug metabolism in vitro, and is not bound to plasma proteins, it is unlikely to produce, or be subject to, pharmacokinetic interactions.
In vivo studies and population pharmacokinetic analysis :
Accordingly, in in vivo studies no clinically relevant pharmacokinetic interactions were observed between pregabalin and phenytoin, carbamazepine, valproic acid, lamotrigine, gabapentin, lorazepam, oxycodone or ethanol. Population pharmacokinetic analysis indicated that oral antidiabetics, diuretics, insulin, phenobarbital, tiagabine and topiramate had no clinically significant effect on pregabalin clearance.
Oral contraceptives, norethisterone and/or ethinyl oestradiol :
Co-administration of pregabalin with the oral contraceptives norethisterone and/or ethinyl oestradiol does not influence the steady-state pharmacokinetics of either substance.
CNS influencing medical products :
Pregabalin may potentiate the effects of ethanol and lorazepam. In controlled clinical trials, multiple oral doses of pregabalin co-administered with oxycodone, lorazepam, or ethanol did not result in clinically important effects on spiration. In the postmarketing experience, there are reports of respiratory failure and coma in patients taking pregabalin and other CNS depressant medicinal products. Pregabalin appears to be additive in the impairment of cognitive and gross motor function caused by oxycodone.
Interactions and the elderly :
No specific pharmacodynamic interaction studies were conducted in elderly volunteers. Interaction studies have only been performed in adults.
SIDE EFFECTS :
Dry mouth, constipation, nausea, vomiting, flatulence; oedema; dizziness, drowsiness, irritability, attention disturbance, disturbances in muscle control and movement, memory impairment, paraesthesia, euphoria, confusion, fatigue, appetite changes, weight gain; changes in sexual function; visual disturbances and ocular disorders (including blurred vision, diplopia, eye strain and eye irritation); less commonly abdominal distension, increased salivation, gastro-oesophageal reflux disease, taste disturbance, thirst, hot flushes, tachycardia, syncope, dyspnoea, chest tightness, nasal dryness, stupor, depersonalisation, depression, insomnia, abnormal dreams, hallucinations, agitation, mood swings, panic attacks, asthenia, speech disorder, dysuria, urinary incontinence, thrombocytopaenia, joint swelling, muscle cramp, myalgia, arthralgia, sweating, and rash; rarely ascites, dysphagia, pancreatitis, hypotension, hypertension, cold extremities, first-degree AV block, arrhythmia, bradycardia, nasopharyngitis, cough, epistaxis, rhinitis, parosmia, pyrexia, rigors, disinhibition, weight loss, hypoglycaemia or hyperglycaemia, renal failure, menstrual disturbances, breast pain, breast discharge, breast hypertrophy, neutropenia, rhabdomyolysis, hyperacusis, hypokalaemia, and leucocytosis; diarrhoea, congestive heart failure, angioedema, loss of consciousness, headache, Stevens-Johnson syndrome, and pruritus also reported.
OVERDOSAGE :
In the post-marketing experience, the most commonly reported adverse reactions observed when pregabalin was taken in overdose included somnolence, confusional state, agitation, and restlessness. In rare occasions, cases of coma have been reported.
TREATMENT OF OVERDOSAGE :
Treatment of pregabalin overdose should include general supportive measures and may include haemodialysis if necessary.
STORAGE :
Store below 30°C (86°F), protected from moisture and light.
Do not refrigerate.
SHELF LIFE :
24 months from date of manufacture.
PRESENTATION :
PREGABALIN CAPSULES I.P. contains Pregabalin I.P.
75 mg / 100 mg / 150 mg / 300 mg.
3 Strips of 10 Capsules per Box.
Disclaimer : For the use of a Registered Medical Practitioner or a Hospital or a Institution only. Also it is not intended to be used by healthcare professionals or patients for the purpose of prescribing or administering these products. Questions regarding the complete and current content of product labeling / specification / presentation should be directed to SGPharma.

 Cardiovascular
Cardiovascular





