1 mg
For the use of a Registered Medical Practitioner or a Hospital or a Institution only.
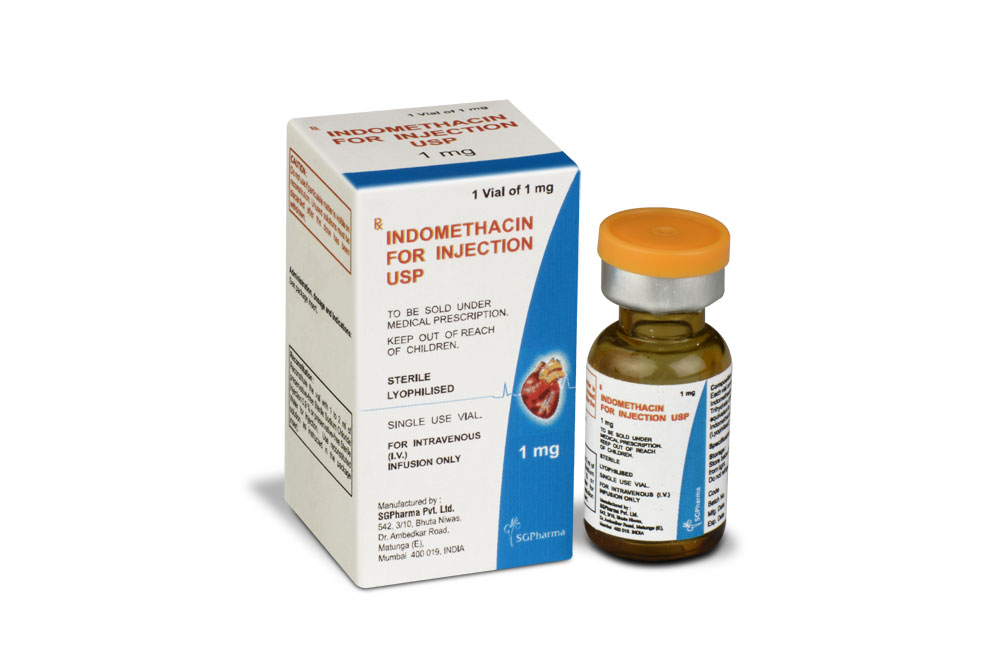
INDOMETHACIN FOR INJECTION USP (Indomethacin) is an indole acetic acid derivative. Indomethacin is used as the sodium salt to close patent ductus arteriosus in premature infants. Chemically, Indomethacin Sodium is 1H -Indole-3-acetic acid, 1-(4-chlorobenzoyl)-5-methoxy-2-methyl-, sodium salt, trihydrate. The molecular formula is C19H15ClNNaO4·3H2O and molecular weight is 433.82.
STRUCTURAL FORMULA :
Its structural formula is :
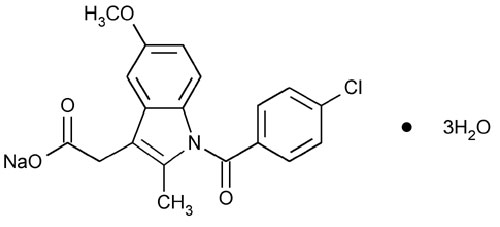
INDOMETHACIN FOR INJECTION USP is a white to slightly yellow lyophilized mass, when reconstituted with water for injection or sterile sodium chloride solution gives a clear and slightly yellow coloured solution.
COMPOSITION :
Each vial contains :
Indomethacin Sodium Trihydrate USP 1.25 mg
equivalent to Indomethacin USP 1.00 mg
(Lyophilized)
CLINICAL PHARMACOLOGY :
Although the exact mechanism of action through which indomethacin causes closure of a patent ductus arteriosus is not known, it is believed to be through inhibition of prostaglandin synthesis. Indomethacin has been shown to be a potent inhibitor of prostaglandin synthesis, both in vitro and in vivo. In human newborns with certain congenital heart malformations, PGE 1 dilates the ductus arteriosus. In foetal and newborn lambs, E type prostaglandins have also been shown to maintain the patency of the ductus, and as in human newborns, indomethacin causes its constriction. Studies in healthy young animals and in premature infants with patent ductus arteriosus indicated that, after the first dose of intravenous indomethacin, there was a transient reduction in cerebral blood flow velocity and cerebral blood flow. Similar decreases in mesenteric blood flow and velocity have been observed. The clinical significance of these effects has not been established. In double-blind placebo-controlled studies of INDOMETHACIN FOR INJECTION USP 460 small pre-term infants, weighing 1750 gm or less, the neonates treated with placebo had a ductus closure rate after 48 hours of 25 to 30 %, whereas those treated with INDOMETHACIN FOR INJECTION USP had a 75 to 80 % closure rate. In one of these studies, a multicenter study, involving 405 pre-term infants, later re-opening of the ductus arteriosus occurred in 26 % of neonates treated with INDOMETHACIN FOR INJECTION USP, however, 70 % of these closed subsequently without the need for surgery or additional indomethacin.
PHARMACOKINETICS :
The disposition of indomethacin following intravenous administration (0.2 mg/kg) in pre-term neonates with patent ductus arteriosus has not been extensively evaluated. Even though the plasma half-life of indomethacin was variable among premature infants, it was shown to vary inversely with postnatal age and weight. In one study, of 28 neonates who could be evaluated, the plasma half-life in those less than 7 days old averaged 20 hours (range : 3 - 60 hours, n = 18). In neonates older than 7 days, the mean plasma half-life of indomethacin was 12 hours (range : 4 - 38 hours, n = 10). Grouping the neonates by weight, mean plasma half-life in those weighing less than 1000 gm was 21 hours (range : 9 - 60 hours, n = 10) ; in those neonates weighing more than 1000 gm, the mean plasma half-life was 15 hours (range : 3 - 52 hours, n = 18). Following intravenous administration in adults, indomethacin is eliminated via renal excretion, metabolism, and biliary excretion. Indomethacin undergoes appreciable enterohepatic circulation. The mean plasma half-life of indomethacin is 4.5 hours. In the absence of enterohepatic circulation, it is 90 minutes. Indomethacin has been found to cross the blood-brain barrier and the placenta. In adults, about 99 % of indomethacin is bound to protein in plasma over the expected range of therapeutic plasma concentrations. The percent bound in neonates has not been studied. In controlled trials in premature infants, however, no evidence of bilirubin displacement has been observed as evidenced by increased incidence of bilirubin encephalopathy (kernicterus).
INDICATIONS :
INDOMETHACIN FOR INJECTION USP is indicated to close a haemodynamically significant patent ductus arteriosus in premature infants weighing between 500 and 1750 gm when after 48 hours usual medical management (e.g., fluid restriction, diuretics, digitalis, respiratory support, etc.) is ineffective. Clear-cut clinical evidence of a haemodynamically significant patent ductus arteriosus should be present, such as respiratory distress, a continuous murmur, a hyperactive precordium, cardiomegaly and pulmonary plethora on chest x-ray.
Administration :
INDOMETHACIN FOR INJECTION USP is for intravenous (I.V.) infusion use only.
Dosage :
Dosage recommendations for closure of the ductus arteriosus depend on the age of the infant at the time of therapy. A course of therapy is defined as three intravenous doses of INDOMETHACIN FOR INJECTION USP given at 12 - 24 hour intervals, with careful attention to urinary output. If anuria or marked oliguria (urinary output < 0.6 ml/kg/hr) is evident at the scheduled time of the second or third dose of INDOMETHACIN FOR INJECTION USP, no additional doses should be given until laboratory studies indicate that renal function has returned to normal.
Dosage according to age is as follows :
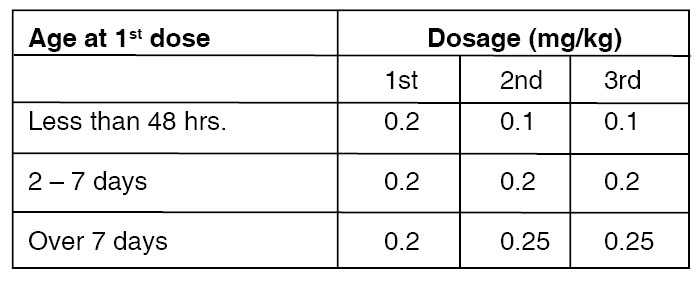
If the ductus arteriosus closes or is significantly reduced in size after an interval of 48 hours or more from completion of the first course of INDOMETHACIN FOR INJECTION USP, no further doses are necessary. If the ductus arteriosus re-opens, a second course of 1 - 3 doses may be given, each dose separated by a 12 - 24 hour interval as described above. If the neonate remains unresponsive to therapy with INDOMETHACIN FOR INJECTION USP after 2 courses, surgery may be necessary for closure of the ductus arteriosus. If severe adverse reactions occur, STOP THE DRUG.
- With proven or suspected infection that is untreated.
- Who are bleeding, especially those with active intracranial haemorrhage or gastrointestinal bleeding.
- With thrombocytopaenia or coagulation defects
- With or who are suspected of having necrotizing enterocolitis
- With significant impairment of renal function
- With congenital heart disease in whom patency of the ductus arteriosus is necessary for satisfactory pulmonary or systemic blood flow (e.g., pulmonary atresia, severe tetralogy of fallot, severe coarctation of the aorta).
WARNINGS :
Gastrointestinal Effects :
In the collaborative study, major gastrointestinal bleeding was no more common in those neonates receiving indomethacin than in those neonates on placebo. However, minor gastrointestinal bleeding (i.e., chemical detection of blood in the stool) was more commonly noted in those neonates treated with indomethacin. Severe gastrointestinal effects have been reported in adults with various arthritic disorders treated chronically with oral indomethacin.
Central Nervous System Effects :
Prematurity per se, is associated with an increased incidence of spontaneous intraventricular haemorrhage. Because indomethacin may inhibit platelet aggregation, the potential for intraventricular bleeding may be increased. However, in the large multicenter study of INDOMETHACIN FOR INJECTION USP, the incidence of intraventricular haemorrhage in neonates treated with INDOMETHACIN FOR INJECTION USP, was not significantly higher than in the control neonates.
Renal Effects :
INDOMETHACIN FOR INJECTION USP may cause significant reduction in urine output (50 % or more) with concomitant elevations of blood urea nitrogen and creatinine, and reductions in glomerular filtration rate and creatinine clearance. These effects in most neonates are transient, disappearing with cessation of therapy with INDOMETHACIN FOR INJECTION USP However, because adequate renal function can depend upon renal prostaglandin synthesis, INDOMETHACIN FOR INJECTION USP may precipitate renal insufficiency, including acute renal failure, especially in neonates with other conditions that may adversely affect renal function (e.g., extracellular volume depletion from any cause, congestive heart failure, sepsis, concomitant use of any nephrotoxic drug, hepatic dysfunction). When significant suppression of urine volume occurs after a dose of INDOMETHACIN FOR INJECTION USP no additional dose should be given until the urine output returns to normal levels. INDOMETHACIN FOR INJECTION USP in pre-term infants may suppress water excretion to a greater extent than sodium excretion. When this occurs, a significant reduction in serum sodium values (i.e., hyponaetremia) may result. Neonates should have serum electrolyte determinations done during therapy with INDOMETHACIN FOR INJECTION USP renal function and serum electrolytes should be monitored.
PRECAUTIONS :
General :
Indomethacin may mask the usual signs and symptoms of infection. Therefore, the physician must be continually on the alert for this and should use the drug with extra care in the presence of existing controlled infection. Severe hepatic reactions have been reported in adults treated chronically with oral indomethacin for arthritic disorders. If clinical signs and symptoms consistent with liver disease develop in the neonate, or if systemic manifestations occur, INDOMETHACIN FOR INJECTION USP should be discontinued. INDOMETHACIN FOR INJECTION USP may inhibit platelet aggregation. In one small study, platelet aggregation was grossly abnormal after indomethacin therapy (given orally to premature infants to close the ductus arteriosus). Platelet aggregation returned to normal by the tenth day. Premature infants should be observed for signs of bleeding. The drug should be administered carefully to avoid extravascular injection or leakage as the solution may be irritating to tissue.
INDOMETHACIN FOR INJECTION USP for I.V. is not approved for use in adults.
INTERACTIONS AND INCOMPATIBILITIES :
Drug interactions :
Since renal function may be reduced by INDOMETHACIN FOR INJECTION USP, consideration should be given to reduction in dosage of those medications that rely on adequate renal function for their elimination. Because the half-life of digitalis (given frequently to pre-term infants with patent ductus arteriosus and associated cardiac failure) may be prolonged when given concomitantly with indomethacin, the neonate should be observed closely; frequent ECGs and serum digitalis levels may be required to prevent or detect digitalis toxicity early. Furthermore, in one study of premature infants treated with INDOMETHACIN FOR INJECTION USP and also receiving either gentamicin or amikacin, both peak and trough levels of these aminoglycosides were significantly elevated. Therapy with indomethacin may blunt the natriuretic effect of furosemide. This response has been attributed to inhibition of prostaglandin synthesis by non-steroidal anti-inflammatory drugs. In a study of 19 premature infants with patent ductus arteriosus treated with either INDOMETHACIN FOR INJECTION USP alone or a combination of INDOMETHACIN FOR INJECTION USP and furosemide, results showed that neonates receiving both INDOMETHACIN FOR INJECTION USP and furosemide had significantly higher urinary output, higher levels of sodium and chloride excretion, and higher glomerular filtration rates than did those receiving INDOMETHACIN FOR INJECTION USP alone. In this study, the data suggested that therapy with furosemide helped to maintain renal function in the premature infant when INDOMETHACIN FOR INJECTION USP was added to the treatment of patent ductus arteriosus.
Indomethacin usually does not influence the hypo-prothrombinaemia produced by anticoagulants. When indomethacin is added to anticoagulants, prothrombin time should be monitored closely. In post marketing experience, bleeding has been reported in patients on concomitant treatment with anticoagulants and INDOMETHACIN FOR INJECTION USP, caution should be exercised when INDOMETHACIN FOR INJECTION USP and anticoagulants are administered concomitantly. In some patients with compromised renal function, the co-administration of an NSAID and an ACE inhibitor or angiotensin II antagonist may result in further deterioration of renal function, including possible acute renal failure, which is usually reversible.
Neonatal Effects :
In rats and mice, oral indomethacin 4.0 mg/kg/day given during the last three days of gestation caused a decrease in maternal weight gain and some maternal and foetal deaths. An increased incidence of neuronal necrosis in the diencephalon in the live-born foetuses was observed. At 2.0 mg/kg/day, no increase in neuronal necrosis was observed as compared to the control groups. Administration of 0.5 or 4.0 mg/kg/day during the first three days of life did not cause an increase in neuronal necrosis at either dose level. Pregnant rats, given 2.0 mg/kg/day and 4.0 mg/kg/day during the last trimester of gestation, delivered offspring whose pulmonary blood vessels were both reduced in number and excessively muscularized. These findings are similar to those observed in the syndrome of persistent pulmonary hypertension of the neonate.
Incompatibilities :
INDOMETHACIN FOR INJECTION USP is reconstituted with preservative – free sodium chloride for injection 0.9 % or preservative-free water for injection. Preparations containing glucose should not be used ; reconstitution at a pH below 6 may cause precipitation of indomethacin. Visual incompatibility has been reported between INDOMETHACIN FOR INJECTION USP and tolazoline hydrochloride, 7.5 and 10 % glucose injection, calcium gluconate, dobutamine, dopamine, cimetidine, gentamycin sulfate, levofloxacin and tobramycin sulfate. A pH below 6 may account for the visual incompatibility of indomethacin sodium and several of these drugs.
SIDE EFFECTS :
In a double-blind, placebo-controlled trial of 405 premature infants weighing less than or equal to 1750 gm with evidence of large ductal shunting, in those neonates treated with indomethacin (n = 206), there was a statistically significantly greater incidence of bleeding problems, including gross or microscopic bleeding into the gastrointestinal tract, oozing from the skin after needle stick, pulmonary haemorrhage, and disseminated intravascular coagulopathy. There was no statistically significant difference between treatment groups in intracranial haemorrhage. The neonates treated with INDOMETHACIN FOR INJECTION USP also had a significantly higher incidence of transient oliguria and elevations of serum creatinine (greater than or equal to 1.8 mg/dl) than did the neonates treated with placebo. The incidences of retrolental fibroplasia (grades III and IV) and pneumothorax in neonates treated with INDOMETHACIN FOR INJECTION USP were no greater than in placebo controls and were statistically significantly lower than in surgically-treated neonates.
The following additional adverse reactions in neonates have been reported from the collaborative study, anecdotal case reports, from other studies using rectal, oral, or intravenous indomethacin for treatment of patent ductus arteriosus or in marketed use. The rates are calculated from a database that contains experience of 849 indomethacin-treated neonates reported in the medical literature, regardless of the route of administration. One year follow up is available on 175 neonates and shows no long-term sequelae which could be attributed to indomethacin. In controlled clinical studies, only electrolyte imbalance and renal dysfunction (of the reactions listed below) occurred statistically significantly more frequently after Indomethacin for Injection than after placebo. Reactions marked with a single asterisk (*) occurred in 3 to 9 % of indomethacin-treated neonates ; those marked with a double asterisk (**) occurred in 3 to 9 % of both indomethacin and placebo-treated neonates. Unmarked reactions occurred in less than 3 percent of neonates.
Renal : Renal failure, renal dysfunction in 41 % of neonates, including one or more of the following : reduced urinary output ; reduced urine sodium, chloride, or potassium, urine osmolality, free water clearance, or glomerular filtration rate ; elevated serum creatinine or BUN ; uremia.
Cardiovascular : Intracranial bleeding**, pulmonary hypertension.
Gastrointestinal : Gastrointestinal bleeding*, vomiting, abdominal distention, transient ileus, gastric perforation, localized perforation(s) of the small and/or large intestine, necrotizing enterocolitis.
Metabolic : Hypo-naetremia*, elevated serum potassium*, reduction in blood sugar, including hypoglycemia, increased weight gain (fluid retention).
Coagulation : Decreased platelet aggregation.
The following adverse reactions have also been reported in neonates treated with indomethacin, however, a causal relationship to therapy with INDOMETHACIN FOR INJECTION USP has not been established :
Cardiovascular : Bradycardia.
Respiratory : Apnea, exacerbation of pre-existing pulmonary infection.
Metabolic : Acidosis/alkalosis.
Hematologic : Disseminated intravascular coagulation, thrombocytopaenia.
Ophthalmic : Retrolental fibroplasia.**
A variety of additional adverse experiences have been reported in adults treated with oral indomethacin for moderate to severe rheumatoid arthritis, osteoarthritis, ankylosing spondylitis, acute painful shoulder and acute gouty arthritis. Their relevance to the pre-term infant receiving indomethacin for patent ductus arteriosus is unknown, however, the possibility exists that these experiences may be associated with the use of INDOMETHACIN FOR INJECTION USP in preterm infants.
INFORMATION FOR PATIENTS :
1.Sterile indomethacin sodium is to be administered intravenously only, over a 5 to 10 second period. Extravasation must be avoided because the solution is irritating to tissues.
2.Restriction of fluid intake (recommended for treatment of premature neonates with a patent ductus arteriosus) should be continued during indomethacin treatment.
3.Administration of 1 mg per kg of body weight (mg/kg) of furosemide immediately following indomethacin has been reported to prevent or reduce indomethacin-induced adverse renal effects without interfering with ductus arteriosus closure. However, furosemide administration is not a generally accepted measure for achieving this purpose. If a significant decrease in renal function occurs following a dose of indomethacin as indicated by a serum creatinine concentration greater than 1.2 to 1.4 mg per dl (106 -124 micromols/l) or other appropriate tests, additional doses should be withheld until urine volume increases to normal levels (i.e., > 1 ml per kg of body weight per hour) and/or laboratory studies indicate return of normal renal function.
4.The medication should be discontinued if any severe adverse reaction, especially hepatic function impairment or disease, occurs.
5.If significant constriction or closure of the ductus arteriosus does not occur following 2 courses (3 doses per course) of indomethacin therapy, surgery may be required.
6.Reopening of the ductus arteriosus may occur following initial closure. Although spontaneous reclosure has occurred in many patients, additional indomethacin or surgery may be required.
OVERDOSAGE AND TREATMENT OF OVERDOSAGE:
Recommended treatment may include :
- Discontinuing or temporarily suspending administration.
- Monitoring the patient and treating observed symptoms. The possibility must be considered that gastrointestinal ulceration or haemorrhage may not occur until several days after administration.
- Haemodialysis is not effective in removing indomethacin from the circulation.

 Cardiovascular
Cardiovascular