75 mg
For the use of a Registered Medical Practitioner or a Hospital or a Institution only.
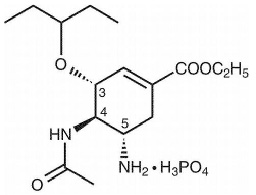
OSELTAMIVIR PHOSPHATE CAPSULES (Oseltamivir Phosphate) is an antiviral agent (influenza). Chemically, Oseltamivir phosphate is (3R,4R,5S)-4-(Acetylamino)-5-amino-3-(1-ethylpropoxy)-1-cyclohexene-1-carboxylic acid ethyl ester, phosphate (1:1). Its molecular formula is C16H28N2O4 (free base) and its molecular weight is 312.40 (as free base) and 410.40 (as phosphate).
STRUCTURAL FORMULA :
Its structural formula is :
OSELTAMIVIR PHOSPHATE CAPSULE is a white to off white crystalline powder filled in capsule of suitable size and colour.
COMPOSITION :
Each hard gelatin capsule contains :
Oseltamivir Phosphate
equivalent to Oseltamivir 75 mg
Excipients q.s.
Approved colours used in empty capsule shells.
ACTIONS :
Oseltamivir phosphate is a pro-drug of oseltamivir carboxylate, a potent and selective inhibitor of influenza virus neuraminidase enzymes. Viral neuraminidase is important both for viral entry into uninfected cells and for the release of recently formed virus particles from infected cells, and the further spread of infectious virus. Oseltamivir carboxylate inhibits the neuraminidases of influenza viruses of both types A and B. Concentrations of oseltamivir carboxylate required to inhibit the enzyme activity by 50 % (IC50) are in the low nanomolar range. Oseltamivir carboxylate also inhibits influenza virus infection and replication in-vitro and inhibits influenza virus replication and pathogenicity in-vivo. Oseltamivir carboxylate reduces shedding of both influenza A and B virus by inhibiting the release of infectious virus from infected cells.
PHARMACOKINETICS :
Absorption :
Oseltamivir is readily absorbed from the gastrointestinal tract after oral administration of oseltamivir phosphate and is extensively converted predominantly by hepatic esterases to the active metabolite. Plasma concentrations of the active metabolite are measurable within 30 minutes, reach near maximal levels in 2 to 3 hours post dose, and substantially exceed (> 20 fold) those of the pro-drug. At least 75 % of an oral dose reaches the systemic circulation as the active metabolite. Plasma concentrations of active metabolite are proportional to dose and are unaffected by co-administration with food.
Distribution :
The mean volume of distribution (VSS) of the active metabolite is approximately 23 litres in humans. The active moiety reaches all key sites of influenza infection as shown by studies in the ferret, rat and rabbit. In these studies, anti-viral concentrations of the active metabolite were seen in the lung, bronchoalveolar lavage, nasal mucosa, middle ear and trachea following oral administration of doses of oseltamivir phosphate. The binding of the active metabolite to human plasma protein is negligible (approximately 3 %). The binding of the pro-drug to human plasma protein is 42 %. These levels are insufficient to cause significant interactions.
Metabolism :
Oseltamivir phosphate is extensively converted to the active metabolite by esterases located predominantly in the liver. Neither oseltamivir nor the active metabolite are substrates for, or inhibitors of, cytochrome P450 isoforms.
Elimination :
Absorbed oseltamivir is primarily (> 90 %) eliminated by conversion to the active metabolite. The active metabolite is not further metabolised and is eliminated in the urine. Peak plasma concentrations of the active metabolite decline with a half-life of 6 to 10 hours in most subjects. The active substance is eliminated entirely (> 99 %) by renal excretion. Renal clearance (18.8 l/h) exceeds glomerular filtration rate (7.5 l/h) indicating that tubular secretion in addition to glomerular filtration occurs. Less than 20 % of an oral radiolabelled dose is eliminated in faeces.
Pharmacokinetics in special populations :
Patients with renal impairment :
Administration of 100 mg of oseltamivir phosphate twice daily for five days to patients with various degrees of renal impairment showed that exposure to the active metabolite is inversely proportional to declining renal function.
Treatment of influenza :
No dose adjustment is necessary for patients with creatinine clearance above 30 ml/min. In patients with a creatinine clearance of 10 - 30 ml/min, it is recommended that the dose be reduced to 75 mg of oseltamivir phosphate once daily for 5 days. No dosing recommendation is available for patients undergoing routine haemodialysis and continuous peritoneal dialysis with end stage renal disease and for patients with creatinine clearance ≤10 ml/min.
Prophylaxis of influenza :
In patients with creatinine clearance between 10 and 30 ml/min receiving oseltamivir phosphate it is recommended that the dose be reduced to 75 mg of oseltamivir phosphate every other day. No dosing recommendation is available for patients undergoing routine haemodialysis and continuous peritoneal dialysis with end stage renal disease and for patients with creatinine clearance ≤10 ml/min.
Patients with hepatic impairment :
In-vitro studies have shown that exposure to oseltamivir is not expected to be increased significantly nor is exposure to the active metabolite significantly decreased in patients with hepatic impairment.
Elderly :Exposure to the active metabolite at steady state was 25 – 35 % higher in elderly (age range 65 - 78) compared to young adults who were given comparable doses of oseltamivir phosphate. Half-lives observed in the elderly were similar to those seen in young adults. On the basis of exposure and tolerability, dosage adjustments are not required for elderly patients for either the treatment or prophylaxis of influenza.
Children :
The pharmacokinetics of oseltamivir phosphate have been evaluated in a single dose pharmacokinetic studies in children aged 1 to 16 years. Multiple dose pharmacokinetics were studied in a small number of children aged 3 - 12 enrolled in a clinical trial. Younger children cleared both the pro-drug and the active metabolite faster than adults resulting in lower exposure for a given mg/kg dose. Doses of 2 mg/kg give oseltamivir carboxylate exposures comparable to those achieved in adults receiving a single 75 mg capsule dose (approximately 1 mg/kg). The pharmacokinetics of oseltamivir in children over 12 years of age are similar to those in adults.
INDICATIONS :
Treatment of Influenza :
OSELTAMIVIR PHOSPHATE CAPSULES is indicated for the treatment of uncomplicated acute illness due to influenza infection in patients 1 year and older who have been symptomatic for not more than 2 days.
Prophylaxis of Influenza :
OSELTAMIVIR PHOSPHATE CAPSULES is indicated for the prophylaxis of influenza in adult patients and adolescents 13 years and older. OSELTAMIVIR PHOSPHATE CAPSULES is not a substitute for early vaccination on an annual basis as recommended by the Centers for Disease Control’s Immunization Practices Advisory Committee.
Administration :
OSELTAMIVIR PHOSPHATE CAPSULES may be taken with or without food. However, when taken with food, tolerability may be enhanced in some patients.
Dosage :
Standard Dosage – Treatment of Influenza :
Adults and Adolescents :
The recommended oral dose of OSELTAMIVIR PHOSPHATE CAPSULES for treatment of influenza in adults and adolescents 13 years and older is 75 mg twice daily for 5 days. Treatment should begin within 2 days of onset of symptoms of influenza.
Paediatric Patients :
OSELTAMIVIR PHOSPHATE CAPSULES is not indicated for treatment of influenza in paediatric patients younger than 1 year.
Standard Dosage – Prophylaxis of Influenza :
Adults and Adolescents :
The recommended oral dose of OSELTAMIVIR PHOSPHATE CAPSULES for prophylaxis of influenza in adults and adolescents 13 years and older following close contact with an infected individual is 75 mg once daily for at least 10 days. Therapy should begin within 2 days of exposure. The recommended dose for prophylaxis during a community outbreak of influenza is 75 mg once daily. Safety and efficacy have been demonstrated for up to 6 weeks. The duration of protection lasts for as long as dosing is continued.
Paediatric Patients :
The safety and efficacy of OSELTAMIVIR PHOSPHATE CAPSULES for prophylaxis of influenza in pediatric patients younger than 1 year of age have not been established.
Special Dosage Instructions :
Hepatic Impairment :
No dose adjustment is recommended for patients with mild or moderate hepatic impairment.
Renal Impairment :
For plasma concentrations of oseltamivir carboxylate predicted to occur following various dosing schedules in patients with renal impairment are seen.
Treatment of Influenza :
Dose adjustment is recommended for patients with creatinine clearance between 10 and 30 ml/min receiving OSELTAMIVIR PHOSPHATE CAPSULES for the treatment of influenza. In these patients it is recommended that the dose be reduced to 75 mg of OSELTAMIVIR PHOSPHATE CAPSULES once daily for 5 days. No recommended dosing regimens are available for patients undergoing routine haemodialysis and continuous peritoneal dialysis treatment with end-stage renal disease.
Prophylaxis of Influenza :
For the prophylaxis of influenza, dose adjustment is recommended for patients with creatinine clearance between 10 and 30 ml/min receiving OSELTAMIVIR PHOSPHATE CAPSULES. In these patients it is recommended that the dose be reduced to 75 mg of OSELTAMIVIR PHOSPHATE CAPSULES every other day or 30 mg every day. No recommended dosing regimens are available for patients undergoing routine haemodialysis and continuous peritoneal dialysis treatment with end-stage renal disease.
Geriatric Patients :
No dose adjustment is required for geriatric patients.
CONTRAINDICATIONS :
OSELTAMIVIR PHOSPHATE CAPSULES are contraindicated in patients with known hypersensitivity to any of the components of the product.
WARNINGS AND PRECAUTIONS :
General :
There is no evidence for efficacy of OSELTAMIVIR PHOSPHATE CAPSULES in any illness caused by agents other than influenza viruses Types A and B. Use of OSELTAMIVIR PHOSPHATE CAPSULES should not affect the evaluation of individuals for annual influenza vaccination in accordance with guidelines of the Centers for Disease Control and Prevention Advisory Committee on Immunization Practices. Efficacy of OSELTAMIVIR PHOSPHATE CAPSULES in patients who begin treatment after 40 hours of symptoms has not been established. Efficacy of OSELTAMIVIR PHOSPHATE CAPSULES in the treatment of subjects with chronic cardiac disease and/or respiratory disease has not been established. No difference in the incidence of complications was observed between the treatment and placebo groups in this population. No information is available regarding treatment of influenza in patients with any medical condition sufficiently severe or unstable to be considered at imminent risk of requiring hospitalization. Safety and efficacy of repeated treatment or prophylaxis courses have not been studied. Efficacy of OSELTAMIVIR PHOSPHATE CAPSULES for treatment or prophylaxis has not been established in immunocompromised patients. Serious bacterial infections may begin with influenza-like symptoms or may coexist with or occur as complications during the course of influenza. OSELTAMIVIR PHOSPHATE CAPSULES has not been shown to prevent such complications.
Hepatic Impairment :
The safety and pharmacokinetics in patients with severe hepatic impairment have not been evaluated.
Renal Impairment :
Dose adjustment is recommended for patients with a serum creatinine clearance < 30 ml/min.
Serious Skin/Hypersensitivity Reactions :
Rare cases of anaphylaxis and serious skin reactions including toxic epidermal necrolysis, Stevens-Johnson Syndrome, and erythema multiforme have been reported. OSELTAMIVIR PHOSPHATE CAPSULES should be stopped and appropriate treatment instituted if an allergic-like reaction occurs or is suspected.
Neuropsychiatric Events :
Influenza can be associated with a variety of neurologic and behavioral symptoms which can include events such as hallucinations, delirium, and abnormal behavior, in some cases resulting in fatal outcomes. These events may occur in the setting of encephalitis or encephalopathy but can occur without obvious severe disease. There have been reports of delirium and abnormal behavior leading to injury, and in some cases resulting in fatal outcomes, in patients with influenza who were receiving OSELTAMIVIR PHOSPHATE CAPSULES. These events were reported primarily among paediatric patients and often had an abrupt onset and rapid resolution. The contribution of OSELTAMIVIR PHOSPHATE CAPSULES to these events has not been established. Patients with influenza should be closely monitored for signs of abnormal behavior. If neuropsychiatric symptoms occur, the risks and benefits of continuing treatment should be evaluated for each patient.
Pregnancy : Category C
There are insufficient human data upon which to base an evaluation of risk of OSELTAMIVIR PHOSPHATE CAPSULES to the pregnant woman or developing foetus. Studies for effects on embryo-foetal development were conducted in rats (50, 250, and 1500 mg/kg/day) and rabbits (50, 150, and 500 mg/kg/day) by the oral route. Relative exposures at these doses were, respectively, 2, 13, and 100 times human exposure in the rat and 4, 8, and 50 times human exposure in the rabbit. Pharmacokinetic studies indicated that foetal exposure was seen in both species. In the rat study, minimal maternal toxicity was reported in the 1500 mg/kg/day group.
In the rabbit study, slight and marked maternal toxicities were observed, respectively, in the 150 and 500 mg/kg/day groups. There was a dose-dependent increase in the incidence rates of a variety of minor skeletal abnormalities and variants in the exposed offspring in these studies. However, the individual incidence rate of each skeletal abnormality or variant remained within the background rates of occurrence in the species studied.
Because animal reproductive studies may not be predictive of human response and there are no adequate and well-controlled studies in pregnant women, OSELTAMIVIR PHOSPHATE CAPSULES should be used during pregnancy only if the potential benefit justifies the potential risk to the foetus.
Nursing Mothers :
In lactating rats, oseltamivir and oseltamivir carboxylate are excreted in the milk. It is not known whether oseltamivir or oseltamivir carboxylate is excreted in human milk. OSELTAMIVIR PHOSPHATE CAPSULES should, therefore, be used only if the potential benefit for the lactating mother justifies the potential risk to the breast-fed infant.
Paediatric Use :
The safety and efficacy of OSELTAMIVIR PHOSPHATE CAPSULES in paediatric patients younger than 1 year of age have not been studied. OSELTAMIVIR PHOSPHATE CAPSULES is not indicated for either treatment or prophylaxis of influenza in paediatric patients younger than 1 year of age because of uncertainties regarding the rate of development of the human blood-brain barrier.
INTERACTIONS AND INCOMPATIBILITIES :
Information derived from pharmacology and pharmacokinetic studies of oseltamivir phosphate suggest that clinically significant interactions with other medicines are unlikely. Oseltamivir phosphate is extensively converted to the active compound by esterases, located predominantly in the liver. Interactions involving competition for esterases have not been extensively reported in the literature. Low protein binding of oseltamivir and the active metabolite do not suggest the probability of displacement interactions. In-vitro studies demonstrated that neither oseltamivir phosphate nor the active metabolite is a good substrate for P450 mixed-function oxidases or for glucuronyl transferases. There is no mechanistic basis for an interaction with oral contraceptives. Cimetidine, a non-specific inhibitor of cytochrome P450 isoforms and competitor for renal tubular secretion of basic or cationic agents has no effect on plasma levels of oseltamivir or its active metabolite.
Clinically important interactions involving competition for renal tubular secretion are unlikely due to the known safety margin for most of these medicines, the elimination characteristics of the active metabolite (glomerular filtration and anionic tubular secretion) and the excretion capacity of these pathways. Co-administration of probenecid results in approximate 2-fold increase in exposure to the active metabolite due to a decrease in active tubular secretion in the kidney. However, due to the wide safety margin of the active metabolite, no dose adjustments are required when co-administering with probenecid. Co-administration with amoxicillin does not alter plasma levels of either compound, indicating that competition for the anionic secretion pathway is weak. Co-administration with paracetamol does not alter plasma levels of oseltamivir, its active metabolite, or paracetamol. No pharmacokinetic interactions between oseltamivir or its major metabolite have been observed when co-administering oseltamivir with paracetamol, acetyl-salicylic acid, cimetidine or with antacids (magnesium and aluminium hydroxides and calcium carbonates).
SIDE EFFECTS :
The most commonly reported adverse effects associated with oseltamivir are nausea and vomiting, abdominal pain, bronchitis, insomnia, and vertigo. Diarrhoea, dizziness, headache, cough, and fatigue may occur, but many adverse effects may be difficult to distinguish from the symptoms of influenza. Other adverse effects occurring less commonly have included unstable angina, anaemia, pseudomembranous colitis, pneumonia, pyrexia, and peritonsillar abscess. There have been occasional reports of skin rash and, rarely, elevated liver enzymes and hepatitis.
INFORMATION FOR PATIENTS :
Patients should be instructed to begin treatment with OSELTAMIVIR PHOSPHATE CAPSULES as soon as possible from the first appearance of flu symptoms. Similarly, prevention should begin as soon as possible after exposure, at the recommendation of a physician.
Patients should be instructed to take any missed doses as soon as they remember, except if it is near the next scheduled dose (within 2 hours), and then continue to take OSELTAMIVIR PHOSPHATE CAPSULES at the usual times. OSELTAMIVIR PHOSPHATE CAPSULES is not a substitute for a flu vaccination. Patients should continue receiving an annual flu vaccination according to guidelines on immunization practices.
OVERDOSAGE :
At present there has been no experience with overdose, however the anticipated manifestations of acute overdose would be nausea, with or without accompanying emesis. Single doses of up to 1000 mg of OSELTAMIVIR PHOSPHATE CAPSULES have been well tolerated apart from nausea and/or vomiting.
TREATMENT OF OVERDOSAGE :
Patients should discontinue the treatment in the event of overdose. No specific antidote is known.
STORAGE :
Protected from moisture and light.
Do not refrigerate.
SHELF LIFE :
24 months from the date of manufacture.
PRESENTATION :
Oseltamivir phosphate Capsules contains Oseltamivir Phosphate equivalent to Oseltamivir 75 mg.
1 Strip of 10 Capsules per Box.

 Cardiovascular
Cardiovascular