3 gm/20 ml
For the use of a Registered Medical Practitioner or a Hospital or a Institution only.
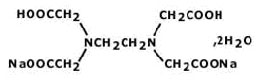
DISODIUM EDETATE INJECTION I.P. (Disodium edetate) is used in emergency treatment of hypercalcaemia and for control of digitalis–induced cardiac arrhythmias. Chemically, Disodium Edetate is disodium ethylenediaminetetraacetate dihydrate. The molecular formula is C10H14N2Na2O8,2H2O and the molecular weight is 372.24.
STRUCTURAL FORMULA :
Its structural formula is :
DISODIUM EDETATE INJECTION I.P. is a sterile, clear, colourless solution filled in vial of suitable size.
COMPOSITION :
Each ml contains :
Disodium Edetate I.P. 150 mg
(anhydrous)
Water for Injections I.P. q.s.
Contains no preservatives.
ACTIONS :
Disodium edetate is classified as a clinical chelating agent for emergency lowering of serum calcium in hypercalcaemia. Disodium edetate forms chelates with the cations of calcium and many divalent and trivalent metals. Because of its affinity for calcium, disodium edetate will produce a lowering of the serum calcium level during intravenous infusion. Slow infusion over a protracted period may cause mobilization of extracirculatory calcium stores. Disodium edetate exerts a negative inotropic effect upon the heart.
PHARMACOKINETICS :
After intravenous administration, the chelate formed is excreted in the urine with 50 % appearing in 1 hour and over 95 % in 24 hours. Disodium edetate likewise forms chelates with other polyvalent metals and produces increases in urinary excretion of magnesium, zinc and other trace elements. It does not form a chelate with potassium but may reduce the serum level and increase urinary loss of potassium.
INDICATIONS :
DISODIUM EDETATE INJECTION i.p. is indicated in selected patients for the emergency treatment of hypercalcaemia and for the control of ventricular arrhythmias associated with digitalis toxicity.
Administration :
FOR I.V. INFUSION AFTER DILUTION
Dosage :
Disodium edetate is given in adult dose of 50 mg/kg in 24 hours by slow intravenous infusion; the maximum daily dose is 3 g. Children may be given 40 to 70 mg/kg in 24 hours. The injection should be diluted with 500 ml of sodium chloride 0.9 % or glucose 5 % for adults or to a concentration not greater than 3 % for children and infused over 3 hours or more, preferably 4 to 6 hours. The dosage may be repeated for a further four-days followed by a two-day interval before subsequent courses of treatment. If necessary, up to fifteen doses may be given in total.
CONTRAINDICATIONS :
DISODIUM EDETATE INJECTION i.p. contraindicated in anuric patients. It is not indicated for the treatment of generalized arteriosclerosis associated with advancing age.
WARNINGS :
Rapid intravenous infusion or attainment of high serum concentration of disodium edetate may cause a precipitous drop in the serum calcium level and many result in fatality. Toxicity appears to be dependent upon both total dosage and speed of administration. The rate of administration and dosage should not exceed that indicated in DOSAGE AND ADMINISTRATION.
PRECAUTIONS :
After the infusion of disodium edetate, the patient should remain in bed for a short time because of the possibility of postural hypotension. The possibility of an adverse effect on myocardial contractility should be considered when administering the drug to patients with heart disease. Caution is dictated in the use of this drug in patients with limited cardiac reserve or incipient congestive failure. DISODIUM EDETATE INJECTION I.P. therapy should be used with caution in patients with clinical or subclinical potassium deficiency states. In such cases it is advisable to perform serum potassium blood levels for possible hypokalemia and to monitor ECG changes. The possibility of hypomagnesemia should be kept in mind during prolonged therapy. Treatment with disodium edetate has been shown to cause a lowering of blood sugar and insulin requirements in patients with diabetes who are treated with insulin.
LABORATORY TEST :
Renal excretory function should be assessed prior to treatment. Periodic BUN and creatinine determinations and daily urinalysis should be performed on patients receiving this drug. Because of the possibility of inducing an electrolyte imbalance during treatment with edetate disodium, appropriate laboratory determinations and studies to evaluate the status of cardiac function should be performed. Repetition of these tests is recommended as often as clinically indicated, particularly in patients with ventricular arrhythmia and those with a history of seizure disorders or intracranial lesions. If clinical evidence suggests any disturbance of liver function during treatment, appropriate laboratory determinations should be performed and withdrawal of the drug may be required.
Pregnancy : Category C
Animal reproduction studies have not been conducted with disodium edetate Injection. It is also not known whether disodium edetate Injection can cause foetal harm when administered to a pregnant woman or can affect reproduction capacity. DISODIUM EDETATE INJECTION i.p. should be given to a pregnant woman only if clearly needed.
Nursing mothers :
The safety of this product in nursing mothers has not been established.
Paediatric Use :
Safety and effectiveness in Paediatric patients have not been established.
Geriatric Use :
Clinical studies of DISODIUM EDETATE INJECTION i.p. did not include sufficient numbers of patients aged 65 and over to determine whether they respond differently from younger subjects. Other reported clinical experience has not identified differences in responses between elderly and younger patients. In general, dose selection for an elderly patient should be cautious, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy. This drug is known to be substantially excreted by the kidney, and the risk of toxic reactions to this drug may be greater in patients with impaired renal function. Because elderly patients are more likely to have decreased renal function, care should be taken in dose selection, and it may be useful to monitor renal function.
Gastrointestinal symptoms such as nausea, vomiting and diarrhoea are fairly common following administration of this drug. Transient symptoms such as circumoral paresthesia, numbness and headache and a transient drop in systolic and diastolic blood pressure may occur. Thrombophlebitis, febrile reactions, hyperuricemia, anemia, exfoliative dermatitis and other toxic skin and mucous membrane reactions have been reported. Nephrotoxicity and damage to the reticuloendothelial system with hemorrhagic tendencies have been reported with excessive dosages.
OVERDOSAGE :
Because of the possibility that DISODIUM EDETATE INJECTION I.P. may produce a precipitous drop in the serum calcium level. A source of calcium replacement suitable for intravenous administration (such as calcium gluconate) should be instantly available at the bedside before edetate disodium is administered. Extreme caution is dictated in the use of intravenous calcium in the treatment of tetany, especially in digitalized patients because the action of the drug and the replacement of calcium ions may produce a reversal of the desired digitalis effect.
TREATMENT OF OVERDOSAGE :
Specific treatment :
STORAGE :
Store below 30°C (86°F), protected from light.
SHELF LIFE :
24 months from the date of manufacture.
Disclaimer : For the use of a Registered Medical Practitioner or a Hospital or a Institution only. Also it is not intended to be used by healthcare professionals or patients for the purpose of prescribing or administering these products. Questions regarding the complete and current content of product labeling / specification / presentation should be directed to SGPharma.

 Cardiovascular
Cardiovascular