600 mg
For the use of a Registered Medical Practitioner or a Hospital or a Institution only.
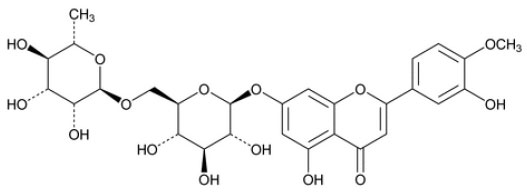
DIOSMIN TABLETS (Diosmin) is vascular-protecting agent. Chemically, Diosmin is 7-[[6-O-(6-Deoxy-α-L-mannopyranosyl)-β-D-glucopyranosyl]oxy]-5-hydroxy-2-(3-hydroxy-4-methoxyphenyl)-4H-1-benzopyran-4-one. The molecular formula is C28H32O15 and molecular weight is 608.54.
STRUCTURAL FORMULA :
Its structural formula is :
DIOSMIN TABLETS are orange coloured, biconvex, film coated tablets with break line on one side.
COMPOSITION :
Each film coated tablet contains :
Diosmin USP 600 mg
Excipients q.s.
Colours : Titanium Dioxide B.P., Tartrazine, Sunset Yellow
ACTIONS :
Diosmin’s mechanisms of action include improvement of venous tone, increased lymphatic drainage, protection of capillary bed microcirculation, inhibition of inflammatory reactions, and reduced capillary permeability. Certain flavonoids, including diosmin, are potent inhibitors of prostaglandin E2 (PGE2) and thromboxane A2 (TxA2) as well as being inhibitors of leucocyte activation, migration, and adhesion. Diosmin causes a significant decrease in plasma levels of endothelial adhesion molecules and reduces neutrophil activation, thus providing protection against microcirculatory damage.
PHARMACOKINETICS :
Pharmacokinetic investigations have shown diosmin is rapidly transformed by intestinal flora to its aglycone form, Diosmetin. Diosmetin is absorbed and rapidly distributed throughout the body with a plasma half-life of 26-43 hours. Diosmetin is degraded to phenolic acids or their glycine-conjugated derivatives and eliminated through the urine. Diosmin or diosmetin not absorbed is eliminated in the faeces.
INDICATIONS :
DIOSMIN TABLETS are indicated for :
• Treatment of symptoms related to venolymphatic insufficiency (heavy legs, pain, early morning restless legs, varicosis, oedema , swelling ),
• Treatment of functional symptoms related to acute haemorrhoidal attack.
• Treatment of Chronic Venous insufficiency (CVI).
• Capillary fragility.
Administration :
DIOSMIN TABLETS are for oral administration only.
Dosage :
• Usual dosage : 2 tablets daily in two divided doses, midday and evening at meal times.
• Acute haemorrhoidal attack : 6 tablets per day for the first 4 days, then 4 tablets per day for 3 days.
• For maintenance in the haemorrhoid illness : 2 tablets to the day.
CONTRAINDICATIONS :
Known hypersensitivity to Diosmin or to any of the excipients.
DIOSMIN TABLETS contains lactose which is contra-indicated in patients with galactosaemia, the glucose-galactose malabsorption syndrome, or lactase deficiency.
Acute haemorrhoidal attack :
Administration of this medicine is no substitute for the specific treatment of other haemorrhoidal disorders. The treatment must be short-term. If the symptoms do not disappear rapidly, proctological examination should be performed and the treatment reviewed.
The tablet contains “tartrazine” colour which may cause allergic reactions, including bronchial asthma, especially in patients who are allergic to the acetyl salicylic acid.
DIOSMIN TABLETS should be used cautiously in diabetic patients.
Pregnancy :
Animal studies showed no teratogenic effect. In the absence of teratogenic effects in animals, malformation in humans is not expected. Indeed, the substance responsible for malformations in the human species was revealed in animals during studies conducted on the two species. In the clinic, no malformations or particular foetal toxicity have appeared to date. However, the outcome of pregnancies exposed to diosmin is insufficient to exclude all risk. Consequently, the necessity of this medicine must be considered during pregnancy.
INTERACTIONS :
Diosmin can cause a decrease in RBC aggregation and blood viscosity. There are no documented cases of adverse interactions between diosmin and prescription medications, but caution should be taken when combining diosmin with aspirin or other blood-thinning medications. Data suggest that diosmin has an inhibitory effect on cytochrome P450-mediated metabolism in healthy volunteers, which may alter the pharmacokinetics of drugs taken concomitantly. Patients given metronidazole after nine days of pretreatment with 450 mg diosmin demonstrated changes in serum concentrations of metronidazole, as well as changes in urinary concentrations of metronidazole and its metabolites compared to control .
SIDE EFFECTS :
Commonly reported adverse events included gastrointestinal disturbances and headaches; these were generally mild in severity and did not usually result in patients discontinuing participation in the study. The following adverse events (and approximate percentages) were reported in some clinical trials but their frequency did not differ from placebo : rash (1 %), cramping in lower limb (2 %), phlebitis (2 %), venous thrombosis (4 %), and skin changes around existing ulcer, swelling of the extremities and body rash (1.6 %).

 Cardiovascular
Cardiovascular