300 mg/5 ml
For the use of a Registered Medical Practitioner or a Hospital or a Institution only.
Fosfestrol (diethylstilboestrol diphosphate tetrasodium salt, DESPP) can be regarded as a water-soluble transport form of the synthetic oestrogen diethylstilboestrol (DES), which is converted to its active forms by enzymatic cleavage of the phosphoric acid groups by acid phosphatases. Chemically, Fosfestrol Sodium is a hydrate of tetrasodium (E)-4,4’-(1,2-diethylvinylene)bis(phenylorthophosphate).The molecular formula is C18H18Na4O8P2. x H2O and the molecular weight is 516.2.
STRUCTURAL FORMULA :
Its structural formula is :
-Structure.jpg)
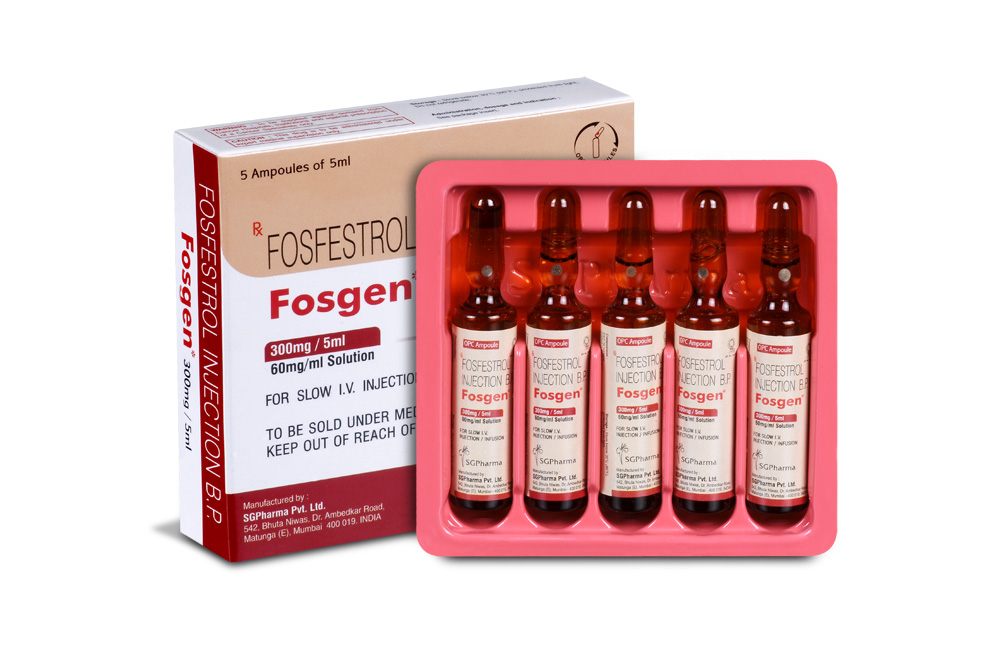
FOSGEN is a clear colourless solution filled in 5 mL amber ampoule.
COMPOSITION :
Each mL contains :
Fosfestrol Sodium B.P. equivalent to
Fosfestrol Sodium anhydrous 60 mg
Water for Injections B.P. q.s.
ACTIONS :
Administration of oestrogens to men leads via negative feedback mechanisms (as yet incompletely understood) in the hypothalamic-hypophyseal-gonadal axis-probably via interactions with androgen receptors – to a reduction in hypophyseal secretion of LH and hence to a reduction in testosterone production. Testosterone concentrations fall to levels seen after castration. In addition there is direct interference with testicular androgen production. Oestrogens also cause a rise in serum levels of prolactin and sex hormone building globulins; this results in a shift towards inactive (bound) testosterone in the serum. Changes also take place in hepatic steroid breakdown, the reticulo-endothelial system and immune status. In addition to their hormonal properties, fosfestrol and its metabolites DES monophosphate and DES have been shown to have marked cytotoxic actions in a variety of tumour tissues. The mean effective dose exerting a direct cytotoxic and antimitotic effect is approximately 0.5 µg/mL of DES. In prostatic tissue DES inhibits DNA polymerase activity and exerts inhibitory effects on DNA and RNA synthesis.
PHARMACOKINETICS :
Metabolism occurs primarily in the liver.
INDICATIONS :
Hormone sensitive as well as hormone resistant metastasizing carcinoma of the prostate.
Administration :
The Injectable solution should be given by slow intravenous injection, the patient being in a supine position. High-dose fosfestrol should be given by infusion (e.g. with glucose or fructose).
INSTRUCTION FOR USE OF AMPOULE :
The ampoule used in this product is equipped with O.P.C (One Point Cut) opening system. No ampoule file is needed to open the ampoule. The neck of the ampoule is prescored at the point of constriction. A coloured dot on the ampoule head helps to orientate the ampoule. Take the ampoule and face the coloured dot. Let the solution at the head of the ampoule to flow down by shaking or a gentle stroke. The ampoule opens easily by placing the thumb on the coloured dot and gently pressing downwards as shown.

Dosage :
There is no fixed regimen that suits every single patient. In any case a distinction should be made between :
1. High-dose initial treatment which may be repeated if required by the patient’s condition.
2. Lower dosed continuous or intermittent maintenance treatment.
A. Initial treatment therapy may begin with daily doses of 1200 mg (= 20 mL = 4 ampoules), the infusion running for about 3 hours. In cases of poor general health and in cases with impaired coronary, heart, liver or kidney function (cf. Contra-indications & precautions) the initial dosage may be reduced down to 600 mg (= 10 mL = 2 ampoules) a day. (Metabolism and way of elimination suggest retarded elimination in case of renal or hepatic malfunction). If tolerated sufficiently well, this starting dosage should be maintained for 10 days.
According to the patient’s response, 10 to 20 days with the dosage reduced to 300 mg (= 5 mL = 1 ampoule) a day may follow. Recent data on bioavailability suggest that suitable plasma levels may also be obtained by oral administration of 360 to 480 mg (3 to 4 tablets) t.i.d. if daily injections or infusions are unfeasible. B. Maintenance treatment : If the initial treatment has resulted in a satisfactory effect, oral maintenance treatment may be started with 120 to 240 mg (1 to 2 tablets) t.i.d. Dosages lower than 2 tablets a day are not recommended. Maintenance treatment may also be carried out with I.V. injections using 300 mg (= 5 mL= 1 ampoule) 1 to 4 times a week, according to the patient’s condition.
Tablets should be taken before meals with little fluid. Sublingual or buccal use is recommended for patients with a sensitive stomach.
CONTRAINDICATIONS :
Unless there are distinct reasons not to use alternative treatment, the side effects profile of fosfestrol suggests to regard as contraindications :
• A history or the presence of thrombophlebitis,thromboembolic disorders, myocardial infarction or cerebral vascular disease.
• Markedly impaired liver function.
WARNINGS AND PRECAUTIONS :
(Dosage reduced, specific observation and specific treatment where possible) is advised in cases of
•Severe atherosclerotic coronary artery obstruction.
•Uncompensated cardiac insufficiency.
•Severe electrolyte imbalance.
•Severe renal impairment.
In diabetics, if not excluded at all (cardiovascular risks), a possible further decrease in glucose tolerance has to be considered. The onset of headaches of a nature not experienced before, visual impairment, neurological disturbances, dyspnoea or mental depression calls for immediate diagnostics and re-evaluation of the treatment with fosfestrol.
DRUG INTERACTIONS :
There are no known specific interactions between fosfestrol and other (non-hormonal) drugs that prostatic carcinoma patients might require. Like always when two or more drugs are given which act in the same direction, untoward side effects may manifest themselves in a more pronounced way when fosfestrol coincides with some other medication able to provoke similar reactions. For example : Enhanced cardiotoxicity is conceivable if fosfestrol is given alongside tricyclic antidepressants. (Enhanced cardiotoxicity has been described for tricyclics in conjunction with oestrogens)
INCOMPATIBILITIES :
Drip solutions containing Calcium or Magnesium salts cannot be used for the infusion of fosfestrol as precipitation may occur.
SIDE EFFECTS :
There are some rather typical adverse reactions that occur mainly during or after injection or infusion. These are :
•Nausea and vomiting
•Burning pain and/or itching in the anogenital region or, on occasion, other parts of the body. Lowered blood pressure has been observed after infusion of very high doses. Other possible side effects may manifest themselves as more slowly developing organic or functional changes, such as
•Thromboembolic complications.
•Coronary and/or cardiac Insufficiency.
•Hepatic manifestations.
•Salt and water retention, oedema.
•Elevated blood pressure.
•Altered serum calcium and phosphate levels.
•Decrease in glucose tolerence.
•Gynecomastia.
•Gastrointestinal complaints (abdominal cramps, bloating and diarrhoea).
•Headaches, aggravation of migraine.
• Mental depression.
•Loss of libido and impaired erectile function.
For the most part, these effects seem to be linked to the oestrogen function. Any other effect attributed to oestrogens in general and apt to express itself in the male also apply for fosfestrol in the present Indication. Like other cytostatics or radiation, fosfestrol itself also possesses a measure of mutagenic and carcinogenic potential, which, however, does not seem to manifest itself within the life span of patients with advanced prostatic carcinoma. Myelosuppression cannot be ruled out, but, unlike with other cytostatics, it seems not to be a typical problem with fosfestrol.
PHARMACEUTICAL PRECAUTIONS :
Parenteral drug products should be inspected visually for particulate matter and discolouration prior to administration, whenever solution and container permit.
STORAGE :
Store below 30°C (86°F), protected from light.
Do not refrigerate.
SHELF LIFE :
24 months from the date of manufacture.
PRESENTATION :
FOSGEN is supplied as 300 mg fosfestrol sodium anhydrous in 5 mL aqueous solution. Such 5 ampoules of 5 mL are packed in a box.
Disclaimer : For the use of a Registered Medical Practitioner or a Hospital or a Institution only. Also it is not intended to be used by healthcare professionals or patients for the purpose of prescribing or administering these products. Questions regarding the complete and current content of product labeling / specification / presentation should be directed to SGPharma.

 Cardiovascular
Cardiovascular