300 mg
For the use of a Registered Medical Practitioner or a Hospital or a Institution only.
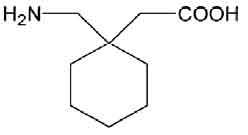
GABAPENTIN CAPSULES USP (Gabapentin) is an anticonvulsant. Chemically, Gabapentin is 1-(Aminomethyl)cyclohexaneacetic acid. The molecular formula is C9H17NO2 and molecular weight is 171.24.
STRUCTURAL FORMULA :
Its structural formula is :
GABAPENTIN CAPSULES USP 300 mg contains white to off white powder filled in hard gelatin capsule of suitable size.
COMPOSITION :
Each hard gelatin capsule contains :
Gabapentin USP 300 mg
Excipients q.s.
Approved colours used in empty capsule shells.
ACTIONS :
The mechanism by which gabapentin exerts its analgesic action is unknown, but in animal models of analgesia, gabapentin prevents allodynia (pain-related behaviour in response to a normally innocuous stimulus) and hyperalgesia (exaggerated response to painful stimuli). In particular, gabapentin prevents pain related responses in several models of neuropathic pain in rats or mice (e.g., spinal nerve ligation models, streptozocin-induced diabetes model, spinal cord injury model, acute herpes zoster infection model). Gabapentin also decreases pain-related responses after peripheral inflammation (carrageenan footpad test, late phase of formalin test). Gabapentin did not alter immediate pain-related behaviours (rat tail flick test, formalin footpad acute phase, acetic acid abdominal constriction
test, footpad heat irradiation test). The relevance of these models to human pain is not known. The mechanism by which gabapentin exerts its anticonvulsant action is unknown, but in animal test systems designed to detect anticonvulsant activity, gabapentin prevents seizures as do other marketed anticonvulsants. Gabapentin exhibits antiseizure activity in mice and rats in both the maximal electroshock and pentylenetetrazole seizure models and other preclinical models (e.g., strains with genetic epilepsy, etc.).
The relevance of these models to human epilepsy is not known. Gabapentin is structurally related to the neurotransmitter GABA (gamma-aminobutyric acid) but it does not modify GABAA or GABAB radioligand binding, it is not converted metabolically into GABA or a GABA agonist, and it is not an inhibitor of GABA uptake or degradation. Gabapentin was tested in radioligand binding assays at concentrations up to 100 µM and did not exhibit affinity for a number of other common receptor sites, including benzodiazepine, glutamate,
N-methyl-D-aspartate (NMDA), quisqualate, kainate, strychnine-insensitive or strychnine-sensitive glycine, alpha 1, alpha 2, or beta adrenergic, adenosine A1 or A2, cholinergic muscarinic or nicotinic, dopamine D1 or D2, histamine H1, serotonin S1 or S2, opiate mu, delta or kappa, cannabinoid 1, voltage-sensitive calcium channel sites labeled with nitrendipine or diltiazem, or at voltage-sensitive sodium channel sites labeled with batrachotoxinin A 20-alpha-benzoate. Furthermore, gabapentin did not alter the cellular uptake of dopamine, noradrenaline, or serotonin. In vitro studies with radiolabeled gabapentin have revealed a gabapentin binding site in areas of rat brain including neocortex and hippocampus. A high-affinity binding protein in animal brain tissue has been identified as an auxiliary subunit of voltage-activated calcium channels. However,functional correlates of gabapentin binding, if any, remain to be elucidated.
PHARMACOKINETICS :
All pharmacological actions following gabapentin administration are due to the activity of the parent compound; gabapentin is not appreciably metabolized in humans.
Oral Bioavailability :
Gabapentin bioavailability is not dose proportional; i.e., as dose is increased, bioavailability decreases. Bioavailability of gabapentin is approximately 60 %, 47 %, 34 %, 33 %, and 27 % following 900, 1200, 2400, 3600, and 4800 mg/day given in 3 divided doses,
respectively. Food has only a slight effect on the rate and extent of absorption of gabapentin (14 % increase in AUC and Cmax).
Distribution :
Less than 3 % of gabapentin circulates bound to plasma protein. The apparent volume of distribution of gabapentin after 150 mg intravenous administration is 58±6 L (mean±SD). In patients with epilepsy, steady-state predose (Cmin) concentrations of gabapentin in cerebrospinal fluid were approximately 20 % of the corresponding plasma concentrations.
Elimination :
Gabapentin is eliminated from the systemic circulation by renal excretion as unchanged drug. Gabapentin is not appreciably metabolized in humans. Gabapentin elimination half-life is 5 to 7 hours and is unaltered by dose or following multiple dosing. Gabapentin elimination rate constant, plasma clearance, and renal clearance are directly proportional to creatinine clearance. In elderly patients, and in patients with impaired renal function, gabapentin plasma clearance is reduced. Gabapentin can be removed from plasma by haemodialysis. Dosage adjustment in patients with compromised renal function or undergoing haemodialysis is recommended.
Special Populations :
Hepatic Disease :
Because gabapentin is not metabolized, no study was performed in patients with hepatic impairment.
Gender :
Although no formal study has been conducted to compare the pharmacokinetics of gabapentin in men and women, it appears that the pharmacokinetic parameters for males and females are similar and there are no significant gender differences.
Race :
Pharmacokinetic differences due to race have not been studied. Because gabapentin is primarily renally excreted and there are no important racial differences in creatinine clearance, pharmacokinetic differences due to race are not expected.
INDICATIONS :
Epilepsy :
Gabapentin is indicated as adjunctive therapy in the treatment of partial seizures with and without secondary generalization in adults and children aged 6 years and above. Gabapentin is indicated as monotherapy in the treatment of partial seizures with and without secondary generalization in adults and adolescents aged 12 years and above.
Treatment of peripheral neuropathic pain :
Gabapentin is indicated for the treatment of peripheral neuropathic pain such as painful diabetic neuropathy and post-herpetic neuralgia in adults.
Administration :
GABAPENTIN CAPSULES USP is for oral administration.
Dosage :
Gabapentin is given orally with or without food. If Gabapentin dose is reduced, discontinued, or substituted with an alternative medication, this should be done gradually over a minimum of 1 week (a longer period may be needed at the discretion of the prescriber).
Postherpetic Neuralgia :
In adults with postherpetic neuralgia, Gabapentin therapy may be initiated as a single 300 mg dose on Day 1, 600 mg/day on Day 2 (divided BID), and 900 mg/day on Day 3 (divided TID). The dose can subsequently be titrated up as needed for pain relief to a daily dose of 1800 mg (divided TID). In clinical studies, efficacy was demonstrated over a range of doses from 1800 mg/day to 3600 mg/day with comparable effects across the dose range. Additional benefit of using doses greater than 1800 mg/day was not demonstrated.
Epilepsy :
Gabapentin is recommended for add-on therapy in patients 3 years of age and older. Effectiveness in paediatric patients below the age of 3 years has not been established. Patients > 12 years of age : The effective dose of Gabapentin is 900 to 1800 mg/day and given in divided doses (three times a day) using 300 or 400 mg capsules, or 600 or 800 mg tablets. The starting dose is 300 mg three times a day. If necessary, the dose may be increased using 300 or 400 mg capsules, or 600 or 800 mg tablets three times a day up to 1800 mg/day. Dosages up to 2400 mg/day have been well tolerated in long-term clinical studies. Doses of 3600 mg/day have also been administered to a small number of patients for a relatively short duration, and have been well tolerated. The maximum time between doses in the TID schedule should not exceed 12 hours.
Paediatric Patients Age 3–12 years : The starting dose should range from 10–15 mg/kg/day in 3 divided doses, and the effective dose reached by upward titration over a period of approximately 3 days. The effective dose of Gabapentin in patients 5 years of age and older is 25–35 mg/kg/day and given in divided doses (three times a day). The effective dose in paediatric patients ages 3 and 4 years is 40 mg/kg/day and given in divided doses (three times a day). Gabapentin may be administered as the oral solution, capsule, or tablet, or using combinations of these formulations. Dosages up to 50 mg/kg/day have been well tolerated in a long-term clinical study. The maximum time interval between doses should not exceed 12 hours. It is not necessary to monitor Gabapentin plasma concentrations to
optimize Gabapentin therapy. Further, because there are no significant pharmacokinetic interactions among Gabapentin and other commonly used antiepileptic drugs, the addition of Gabapentin does not alter the plasma levels of these drugs appreciably. If Gabapentin is discontinued and/or an alternate anticonvulsant medication is added to the therapy, this should be done gradually over a minimum of 1 week.
Dosage in Renal Impairment :
Creatinine clearance is difficult to measure in outpatients. In patients with stable renal function, creatinine clearance (Ccr) can be reasonably well estimated using the equation of Cockcroft and Gault : for females Ccr =(0.85)(140-age)(weight)/[(72)(Scr)] for males Ccr =(140-age)(weight)/[(72)(Scr)] in which age is in years, weight is in kilograms and Scr is serum creatinine in mg/dl.
Dosage in Elderly :
Because elderly patients are more likely to have decreased renal function, care should be taken in dose selection, and dose should be adjusted based on creatinine clearance values in these patients.
CONTRAINDICATION :
GABAPENTIN CAPSULES USP is contraindicated in patients who have demonstrated hypersensitivity to gabapentin or to any of the components of the formulation.
WARNINGS AND PRECAUTIONS :
Suicidal ideation and behaviour have been reported in patients treated with anti-epileptic agents in several indications. A meta-analysis of randomised placebo controlled trials of anti-epileptic drugs has also shown a small increased risk of suicidal ideation and behaviour. The mechanism of this risk is not known and the available data do not exclude the possibility of an increased risk for gabapentin. Therefore patients should be monitored for signs of suicidal ideation and behaviours and appropriate treatment should be
considered. Patients (and caregivers of patients) should be advised to seek medical advice should signs of suicidal ideation or behaviour emerge. If a patient develops acute pancreatitis under treatment with gabapentin, discontinuation of gabapentin should be considered. Although there is no evidence of rebound seizures with gabapentin, abrupt withdrawal of anticonvulsant agents in epileptic patients may precipitate status epilepticus. As with other antiepileptic medicinal products, some patients may experience an increase in seizure frequency or the onset of new types of seizures with gabapentin. As with other anti-epileptics, attempts to withdraw concomitant anti-epileptics in treatment refractive patients on more than one anti-epileptic, in order to reach gabapentin monotherapy have a low success rate.
Gabapentin is not considered effective against primary generalized seizures such as absences and may aggravate these seizures in some patients. Therefore, gabapentin should be used with caution in patients with mixed seizures including absences. No systematic studies in patients 65 years or older have been conducted with gabapentin. In one double blind study in patients with neuropathic pain, somnolence, peripheral oedema and asthenia occurred in a somewhat higher percentage in patients aged 65 years or above, than in younger patients. Apart from these findings, clinical investigations in this age group do not indicate an adverse event profile different from that observed in younger patients. The effects of long-term (greater than 36 weeks) gabapentin therapy on learning, intelligence, and development in children and adolescents have not been adequately studied. The benefits of prolonged therapy must therefore be weighed against the potential risks of such therapy.
Drug Rash with Eosinophilia and Systemic Symptoms (DRESS) :
Severe, life-threatening, systemic hypersensitivity reactions such as Drug rash with eosinophilia and systemic symptoms (DRESS) have been reported in patients taking antiepileptic drugs including gabapentin. It is important to note that early manifestations of
hypersensitivity, such as fever or lymphadenopathy, may be present even though rash is not evident. If such signs or symptoms are present, the patient should be evaluated immediately. Gabapentin should be discontinued if an alternative etiology for the signs or
symptoms cannot be established.
Laboratory tests :
False positive readings may be obtained in the semi-quantitative determination of total urine protein by dipstick tests. It is therefore recommended to verify such a positive dipstick test result by methods based on a different analytical principle such as the Biuret method,
turbidimetric or dye-binding methods, or to use these alternative methods from the beginning.
Paediatric Use :
EFFECTS ON ABILITY TO DRIVE AND USE MACHINES :
undesirable effects could be potentially dangerous in patients driving or operating machinery. This is especially true at the beginning of the treatment and after increase in dose.
INTERACTIONS :
SIDE EFFECTS :
INFORMATION FOR PATIENTS :
Signs and Symptoms
Treatment of Overdosage :
STORAGE :
SHELF LIFE :
PRESENTATION :

 Cardiovascular
Cardiovascular